All published articles of this journal are available on ScienceDirect.
Frailty Characteristics in Chronic HIV Patients are Markers of White Matter Atrophy Independently of Age and Depressive Symptoms: A Pilot Study
Abstract
Background:
Chronic HIV disease is associated with neurocognitive impairment and age-related conditions such as frailty.
Objective:
To determine whether regional brain volumetric changes correlate with frailty parameters in older (≥ 40 years) HIV+ patients on stable combination antiretroviral therapy.
Method:
Thirty-five HIV-infected participants in the Hawaii Aging with HIV Cohort - Cardiovascular Disease study underwent T1-weighted brain magnetic resonance imaging, frailty assessment and neuropsychological testing. Five physical frailty traits were assessed: low physical activity; exhaustion; unintentional weight loss; weak hand grip strength; slow walking speed. Linear regression quantified cross-sectional relationships of 12 brain regions to walking times and hand grip strength.
Results:
Participants were 50.6 ± 6.8 years old and 77% had undetectable plasma viral load. One subject was frail (possessing ≥ 3 frailty traits); 23% were pre-frail (1–2 frailty traits) and had worse composite learning and memory z-scores than did non-frail individuals (p=0.06). Pre-frail or frail subjects had reduced hand grip strength relative to the non-frail group (p=0.001). Longer walking times (slower gait) related independently to lower volumes of cerebellar white matter (p<0.001, β=−0.6) and subcortical gray matter (p<0.05, β=−0.30). Reduced thalamus volume was linked to weaker grip strength (p < 0.05, β=0.4). Caudate volume was negatively associated with grip strength (p<0.01, β=−0.5).
Conclusion:
Volumetric changes in cerebellar white matter and subcortical gray matter, brain regions involved in motor control and cognition, may be connected to frailty development in well-controlled HIV. Gait speed is particularly sensitive to white matter alterations and should be investigated as a predictor of frailty and brain atrophy in chronically infected patients.
INTRODUCTION
The number of older Americans living with human immunodeficiency virus-1 (HIV) has increased greatly in recent years through improved treatment. Among patients on stable combination antiretroviral therapy (cART), HIV has evolved into a chronic disease [1]: by 2020, 70% of HIV-seropositive (HIV+) individuals in the U.S. will be 50 years of age or older [2]. This increased longevity is accompanied by complications that present challenges related to both aging and HIV. HIV-infected patients, even while stable on cART, appear to be aging prematurely and are at increased risk of age-related co-morbidities such as diabetes, cardiovascular disease, and frailty [3]. The life expectancy of cART-treated HIV patients remains 10-30 years below that of the HIV-uninfected population [4]. Decreased lifespan is associated with factors prevalent among HIV+ individuals, such as illicit drug use [5] and co-infection with hepatitis B or C [6].
Frailty is a clinical syndrome associated with falls, cardiovascular disease, disability, institutionalization, and death [7]. It is the eventual result of age-related declines in physiologic reserve and function that lead to a diminished ability to cope with minor stressors [8]. Depressive symptoms are strongly associated with frailty [8]. Characterized by decreases in energy, weight, and physical activity, frailty is more likely to develop in HIV-infected than in HIV-uninfected individuals, and at younger ages [9]. In HIV it has been linked to chronic inflammation [10], immune system deterioration [11], depressive symptoms [12] and HIV-associated neurocognitive disorder (HAND) [13]. HAND occurs in three forms: asymptomatic neurocognitive impairment (mild deficits in at least 2 cognitive areas, which do not affect daily functioning and are apparent only upon neuropsychological testing); mild neurocognitive disorder (mild deficits in 2 or more neuropsychological domains, causing at least mild functional impairment); and HIV-associated dementia (moderate to severe deficits in ≥ 2 areas, with substantially impaired everyday functioning) [14]. Although HAND and HIV disease severity are associated with brain atrophy [15-19], the brain anatomical correlates of frail or pre-frail conditions have not been examined in HIV patients. Our pilot study aimed to determine associations between regional brain volumes and continuous measures of frailty (walking speed and grip strength) in HIV patients on cART. In particular, we evaluated the predictive value of these frailty-related characteristics for brain structural change in chronic HIV infection. We also considered the possibility that regional brain atrophy may be a causative factor in frailty development.
MATERIALS AND METHODS
Subjects
We studied 35 HIV+ individuals who were enrolled in the Hawaii Aging with HIV Cohort - Cardiovascular Disease (HAHC-CVD) study and had complete frailty and brain magnetic resonance imaging (MRI) data. Entry criteria required that subjects be ≥ 40 years old, HIV-seropositive, on stable cART for ≥ 3 months prior to entry, and speak English as their primary language in order to understand and sign a written informed consent document. Exclusion criteria were: (i) uncontrolled major affective disorder; (ii) active psychosis; (iii) loss of consciousness > 5 minutes; (iv) pregnancy or breast-feeding; (v) factors that would preclude MRI (e.g., claustrophobia); and (vi) any past or present condition (central nervous system infection, brain trauma, stroke, substance abuse, etc.) deemed by the evaluating physician to introduce confounding variables. All study participants underwent standard clinical tests, a neurological exam, and neuropsychological (NP) testing. Each subject was assessed for frailty once during the longitudinal study (in Years 2 - 4) and underwent neuroimaging once at Year 2. MRI and frailty testing were thus done approximately 0 - 2 years apart. Blood specimens were obtained and stored at study entry. Plasma HIV RNA (viral load) and CD4 cell counts were performed by a local commercial CLIA-certified laboratory. Nadir CD4 count and date of first HIV diagnosis (used to compute years since HIV diagnosis) were provided by subject self-report or medical records. Racial information was self-reported. Written informed consent was obtained from each participant. The Committee on Human Subjects at the University of Hawaii approved the study.
Neuropsychological Testing
Subjects completed a comprehensive 80-minute neuropsychological test battery that assessed cognitive domains affected by HIV. It included the following tests: Choice and Sequential Reaction Time from the California Computerized Assessment Package (CalCAP); the Rey Auditory Verbal Learning Test (RAVLT); Rey Osterreith Complex Figure (RCF) Copy and Recall; Trail Making Test, Parts A and B; Wechsler Adult Intelligence Scale–Revised (WAIS-R) Digit Symbol; WAIS-III Letter-Number Sequencing; Grooved Pegboard (dominant and nondominant hands); Verbal Fluency Test (FAS); Animal Naming; Boston Naming Test (BNT), the WAIS-R Digit Span (Forward and Backward); Stroop Color Word Interference Test, condition 3; and Timed Gait. Raw NP test scores were z-transformed by use of standard demographically adjusted normative data. Composite, domain-specific z-scores (psychomotor speed [NPZpm]; learning and memory [NPZlrn-mem]; executive functioning [NPZef]; working memory [NPZwm]) were derived by averaging z-scores of the appropriate individual tests. A global z-score (NPZglobal) was the arithmetic mean of z-scores on 14 NP tests. Diagnoses of HAND were not made in this study. Symptoms of depression were assessed using the Beck Depression Inventory (BDI) [20], which was divided into somatic (BDI-S) and cognitive/affective (BDI-CA) subscales according to the procedure described in the BDI manual [21]. BDI somatic and cognitive/affective factors have been previously examined in the context of HIV infection [22]. A total score, BDI-TOT, was obtained.
Physical Frailty
Physical frailty was evaluated using a widely accepted categorical method described by Fried and colleagues [23]. Subjects were assessed for 5 frailty traits: slow gait, decreased grip strength (weakness), poor endurance (exhaustion), low physical activity, and unintentional weight loss of ≥10 lb or ≥ 5% of subject’s body weight since the last visit. Frailty was defined by the presence of ≥ 3 traits; a pre-frail condition defined by 1–2 traits; and non-frailty by 0 traits [23].
Three criteria for frailty (low physical activity, exhaustion, and unintentional weight loss) were self-reported in response to a questionnaire. Subjects were categorized as having a low physical activity level if they said that their health limited them “a lot” in engaging in vigorous activities such as running, strenuous sports, or lifting heavy objects. Evidence of exhaustion was determined by the question “How often in the last week did you feel that (a) that everything you did was an effort or (b) you could not ‘get going’?” Subjects who replied “occasionally” (3-4 days) or “most of the time” (5-7 days) were judged to have poor endurance.
Gait speed was assessed by the average time in seconds to walk 15 feet, computed from two timed walks over a 15-ft course at the subject’s usual pace. Walking times were adjusted for gender and height: 1) men who were <173 cm tall and women <159 cm tall who walked 15 feet in >7 seconds, and 2) men >173 cm in height and women >159 cm who walked 15 feet in >6 seconds, met the frailty criterion of slow gait [23].
Grip strength of the dominant hand was measured 3 times with a Jamar hydraulic hand dynamometer (Model 5030J1, Sammons Preston Rolyan, Inc., Bolingbrook IL). The maximum value was noted. Average grip strength, based on the 3 trials, was used to assess weakness, and the values were adjusted for gender and body mass index (BMI). The criterion for weakness (reduced grip strength) was met by i) men with: BMI ≤ 24 kg/m2 and grip strength ≤ 29 kg; BMI of 24.1 – 28.0 kg/m2 and grip strength ≤ 30 kg; BMI >28 kg/m2 and grip strength ≤ 32 kg; and by ii) women with: BMI ≤ 23 kg/m2 and grip strength ≤ 17 kg; BMI of 23.1 – 26.0 kg/m2 and grip strength ≤ 17.3 kg; BMI of 26.1 – 29.0 kg/m2 and grip strength ≤ 18 kg; and BMI > 29 kg/m2 and grip strength ≤ 21 kg [23]. Regression analyses used both the maximum and average hand grip strength values.
Neuroimaging
Each study participant underwent brain MRI at a 3.0-Tesla Philips Medical Systems Achieva scanner equipped with an 8-channel head coil (InVision Imaging, Honolulu). A high-resolution anatomical volume was acquired with a sagittal T1-weighted 3D turbo field echo (T1W 3D TFE) sequence (echo time TE/repetition time TR=3.2 ms/6.9 ms; flip angle 8°; slice thickness 1.2 mm with no gap; in-plane resolution 1.0 mm2; field of view 256 x 256 mm2). To obtain regional brain volumes, MRI data were processed with FreeSurfer (http://surfer.nmr.mgh.harvard.edu/, version 5.0) [24-26] in a procedure that includes intensity normalization [27], Talairach transformation, subcortical white matter (WM) and deep gray matter (GM) segmentation [25], and cortical gray/white matter boundary and pial surface reconstruction [24]. Quality assurance of processed data was performed visually. Cortical surfaces and subcortical segmentations were checked prior to statistical analysis. FreeSurfer’s estimate of intracranial volume (ICV) can be reliably used to normalize regional brain volumes [28].
Statistical Analyses
Mann-Whitney test and Fisher’s exact test were used as appropriate to assess differences in demographic and clinical variables between non-frail and frail-risk groups, where the frail-risk group consisted of all subjects categorized as frail or pre-frail. Analyses were also done for non-frail vs. pre-frail groups. Total volumes of 12 brain regions (caudate, putamen, pallidum, thalamus, hippocampus, amygdala, nucleus accumbens, cerebellar WM, cerebellar GM, cortical GM, cerebral WM, and total subcortical GM) were obtained by summing over left and right hemispheres. These regions have been found to be affected by HIV [15-19]. Group differences in BDI scores, NP z-scores and regional brain volumes were evaluated by Mann-Whitney test.
Pearson correlation assessed the relationships of continuous frailty-related parameters (walking time, average hand grip strength, and maximum grip strength) to current and nadir CD4 count. Associations between the continuous frailty variables and NP z-scores were examined by Pearson correlation and by multivariate linear regression that adjusted for nadir CD4. Walking times were log-transformed to achieve normal distributions. Pearson correlation was conducted between regional brain volumes and BDI scores (BDI-S, BDI-CA, BDI-TOT), and between BDI scores and continuous frailty parameters. Multivariate regression controlling for ICV also assessed relationships between the volumes (dependent variables) and BDI scores.
A primary objective was to identify the brain regions associated with weak hand grip strength and decreased gait speed, two characteristics of frail and pre-frail conditions. Relationships of regional brain volumes to grip strength and gait speed were first assessed by Pearson correlation and further explored in two types of multivariate regression analyses, distinguished here by the labels “predictive” and “explanatory” notwithstanding the cross-sectional nature of the data. The former type viewed frailty-related characteristics as predictive of changes in regional brain volumes, while explanatory modeling regarded regional brain atrophy as a possible causal factor in the slowing of gait speed and the weakening of hand grip strength.
Slow gait is known to predict cognitive decline [29], which is associated with brain atrophy in HIV infection [30] but also in normal aging [31]. Our predictive regression models used the continuous frailty-related parameters (separately) as independent variables and regional brain volumes as outcomes. Covariates included age, ICV, nadir CD4 count and BDI-TOT. The effects of BDI-S and BDI-CA were checked. For reasons of sample size, nadir CD4 and BDI scores were not included simultaneously in regression analyses.
To address the possibility that brain atrophy may lead to physical frailty, explanatory regression analyses defined continuous frailty parameters as the dependent variables. Independent variables were the regional brain volumes (individually) and age. Use of ICV as a covariate was precluded by collinearity with the volumes. Effects of BDI-S, BDI-CA and BDI-TOT were included when appropriate; i.e., when Pearson correlation showed the BDI score to be related to the dependent variable.
A two-sided p-value <0.05 was considered statistically significant. Trends were defined by 0.05 ≤ p < 0.1. Values of continuous variables are presented as mean ± standard deviation unless otherwise stated. Analyses were conducted in SPSS (IBM, Version 22), Armonk, New York.
RESULTS
Subject Characteristics
This cross-sectional analysis involved 35 HIV+ patients (aged 50.6 ± 6.8 years; 89% male) from the HAHC-CVD cohort. Table 1 presents their demographic, clinical, frailty and neuropsychological characteristics. The study population had a median CD4 cell count of 439.0 cells/mm3 (range: 33.0 – 955.0), a median nadir CD4 count of 87.5 cells/mm3 (range: 0 – 550), and 27 participants (77%) had undetectable plasma HIV RNA (< 50 copies/mL). Subjects had a median BDI score of 8 (range: 0 – 28) and mild global cognitive impairment, with deficits primarily in the domains of learning and memory (NPZlrn-mem= −0.26 ± 1.01) and working memory (NPZwm= −0.32 ± 0.75).
By self-report, 5 study participants had limited physical activity, 2 experienced frequent exhaustion, and 1 had undergone unintentional weight loss. The subjects took 3.5 ± 0.6 seconds to walk 15 feet, and possessed an average grip strength of 36.8 ± 9.0 kg and a maximum grip strength of 38.8 ± 9.3 kg. Walking time was not obtained for one individual who was missing a limb and used a cane. One patient (3% of the sample) was classified as frail (having ≥ 3 frailty traits out of 5) and 8 (23%) were pre-frail (possessing 1–2 frailty traits) [23]. The individual who met criteria for frailty was a Caucasian male, 59 years old, and had undetectable plasma HIV RNA; current and nadir CD4 counts of 273.0 cells/mm3 and 8.0 cells/mm3, respectively; BDI-CA=4, BDI-S=10, BDI-TOT=14; NPZglobal= −0.24, NPZlrn-mem=1.04, NPZpm=0.31, NPZef=0.80, and NPZwm= −1.03. Characteristics of the 8 pre-frail subjects were as follows: 48.9 ± 5.0 years old, 6 (75%) male; 8 (100%) with undetectable HIV RNA; median current CD4 count=467.0 cells/mm3 (range: 116 - 955); median nadir CD4 count=83.0 cells/mm3 (range: 3 – 350); median BDI-CA=7.0 (range: 0 – 19); median BDI-S=4.0 (range: 1–9); median BDI-TOT=10.0 (range: 4 – 28); NPZglobal= −0.18 ± 0.64; NPZlrn-mem= −0.94 ± 0.92; NPZpm=0.38 ± 0.60; NPZef=0.25 ± 0.87; and NPZwm= −0.52 ± 0.42. BDI data were unavailable for one pre-frail study participant.
| Characteristic | All subjects | Non-frail | Frail-risk | p |
|---|---|---|---|---|
| N | 35 | 26 | 9 | – |
| Gender (male) (%) | 31 (89%) | 24 (92%) | 7 (78%) | 0.27 |
| Age (years) | 50.6 ± 6.8 | 50.9 ± 7.4 | 50.0 ± 5.7 | 0.89 |
| Race (Caucasian) (%) | 17 (47%) | 14 (54%) | 3 (33%) | 0.44 |
| Education (years) a | 14.2 ± 1.8 | 14.4 ± 1.9 | 13.5 ± 1.4 | 0.22 |
| Current CD4 count(cells/mm3) | 423.4 ± 192.9 | 415.9 ± 168 | 445 ± 263.2 | 0.96 |
| Nadir CD4 count(cells/mm3) | 139.7 ± 139.6 | 145.3 ± 144.1 | 121.5 ± 130.1 | 0.71 |
| Years since HIV diagnosis | 15.3 ± 7.3 | 15.6 ± 7.5 | 14.4 ± 7.5 | 0.75 |
| # with undetectable HIV RNA (< 50 copies/mL) (%) | 27 (77%) | 18 (69%) | 9 (100%) | 0.08 |
| Average grip strength (kg) | 36.8 ± 9.0 | 39.9 ± 7.3 | 28.0 ± 8.4 | 0.001 |
| Maximum grip strength (kg) | 38.8 ± 9.3 | 42.0 ± 7.6 | 29.3 ± 8.6 | 0.001 |
| Average walking time (sec) a | 3.5 ± 0.6 | 3.4 ± 0.5 | 3.8 ± 0.6 | 0.06 |
| # with unintentional weight loss | 1 | 0 | 1 | 0.26 |
| # with exhaustion | 2 | 0 | 2 | 0.06 |
| # with limited physical activity | 5 | 0 | 5 | 0.0004 |
| Total BDI, median (range)a | 8.0 (0 – 28) | 6.0 (0 – 27) | 11.0 (4 – 28) | 0.09 |
| BDI-cognitive-affective, median (range) a | 4.5 (0 – 19) | 2.5 (0 – 17) | 6.0 (0 – 19) | 0.24 |
| BDI-somatic, median (range) a | 3.0 (0 – 12) | 3.0 (0 – 12) | 5.5 (1 – 10) | 0.13 |
| # with depression (BDI ≥ 20) a | 3 (9%) | 2 (8%) | 1 (13%) | 1.00 |
| NPZglobalb | −0.08 ± 0.53 | −0.06 ± 0.53 | -0.13 ± 0.61 | 0.77 |
| NPZlrn-mem c | −0.26 ± 1.01 | −0.12 ± 0.19 | -0.69 ± 1.10 | 0.19 |
| NPZpmc | 0.33 ± 0.55 | 0.32 ± 0.58 | 0.37 ± 0.55 | 0.73 |
| NPZef a | 0.13 ± 0.92 | 0.07 ± 0.97 | 0.32 ± 0.83 | 0.56 |
| NPZwma | −0.32 ± 0.75 | −0.24 ± 0.83 | -0.58 ± 0.43 | 0.22 |
Structural MRI yielded volumes (mm3) of the thalamus (12995.5 ± 1189.1), caudate (7337.3 ± 887.1), putamen (11421.0 ± 1430.4), pallidum (3295.5 ± 400.9), hippocampus (7950.2 ± 702.5), amygdala (3832.1 ± 528.1), nucleus accumbens (1328.5 ± 218.1), cerebellar white matter (31081.8 ± 3691.6), cerebellar gray matter (88129.5 ± 8433.6), cortical gray matter (436369.8 ± 32303.8 mm3), cerebral white matter (494774.9 ± 58940.8), and subcortical gray matter (165366.5 ± 13605.4). Intracranial volume, a covariate in the statistical analyses, was 1465.8 ± 212.3 cm3.
Group Differences Between Frail-Risk and Non-Frail HIV-Infected Participants
The frail-risk and non-frail groups did not differ significantly in demographic or clinical characteristics (Table 1). All frail-risk individuals but only 69% of non-frail subjects were virally suppressed (p=0.08). Relative to the non-frail group, frail-risk HIV patients had weaker average and maximum hand grip strength (p=0.001 for both). Longer walking times in the frail-risk group were marginally significant (p=0.06). A greater proportion of the frail-risk group suffered from exhaustion (poor endurance) and limitations in physical activity (by definition, these characteristics were not present in non-frail individuals). Regional brain volumes, NP z-scores and the proportion of depressed subjects did not differ between the frail-risk and non-frail groups. Frail-risk subjects exhibited a trend toward higher median BDI-TOT (11.0 vs. 6.0; p=0.09) and had greater BDI somatic and cognitive/affective symptom scores as well, though the differences were too small to constitute a trend.
When group comparisons were performed for pre-frail vs. non-frail subjects (i.e., when the frail patient was excluded from the frail-risk group), p-values in general changed very little. However, the group difference in NPZlrn-mem became borderline significant (p=0.06); there was no trend-level difference in BDI-TOT (p=0.16); and the difference in walking time was non-significant (non-frail: 3.4 ± 0.5; pre-frail: 3.7 ± 0.6; p=0.15).
Relationships of Continuous Frailty Parameters to Age, Current and Nadir CD4 Count, Years Since HIV Diagnosis, Neuropsychological Function, and Depressive Symptoms
We found no significant relationship between any continuous frailty variable (walking time, average grip strength, maximum grip strength) and age, current or nadir CD4 count, years since HIV diagnosis, composite domain-specific NP z-scores, or NPZglobal by Pearson correlation. Multivariate regression models of global and composite NP z-scores against the same frailty measures, adjusting for nadir CD4, showed no associations between NP function and walking speed or grip strength. Total and component BDI scores did not correlate with average or maximum hand grip strength. Longer walking time correlated with more severe depressive symptoms as measured by higher BDI-TOT (p=0.006, R=0.47), BDI-CA (p=0.006, R=0.47), and BDI-S (p=0.061, R=0.33).
Associations Between BDI Scores and Regional Brain Volumes
Pearson correlation revealed a relationship between greater BDI-CA and reduced caudate volume (p=0.023, R= −0.39). No regional volumes correlated significantly with BDI-TOT or BDI-S, but higher values of both scores showed a trend correlation with smaller hippocampal volume (BDI-TOT: p=0.075, R= −0.31; BDI-S: p=0.082, R= −0.30). Linear regression adjusting for ICV found negative relationships between total BDI score and volumes of caudate (p=0.046, β= −0.30) and hippocampus (p=0.053, β= −0.32). Lower caudate volume was associated with higher BDI-CA (p=0.025, R= −0.34). BDI-S was linked to hippocampal (p=0.016, β = −0.40) and cerebellar gray matter (p=0.047, β = −0.33) volumes, and at trend-level with volume of subcortical gray matter (p=0.056, β = −0.28).
Associations Between Continuous Frailty Parameters and Regional Brain Volumes
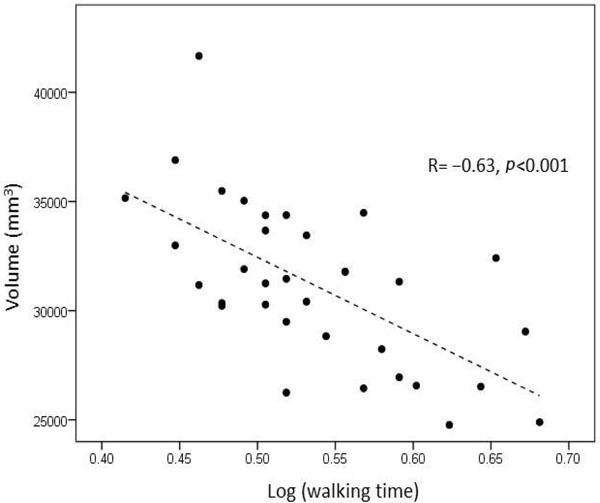
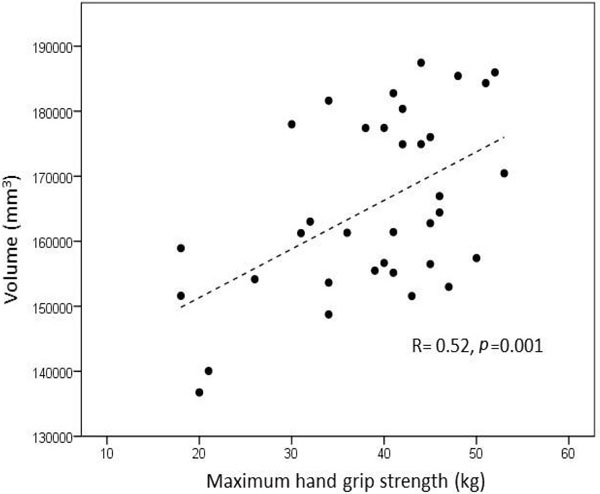
Pearson correlation showed that volumes of selected brain regions were related to gait speed and hand grip strength (Table 2). As shown in Fig. (1), smaller cerebellar WM volume correlated with increased walking time (p<0.001, R= −0.63). At p<0.01, subcortical GM, cerebellar GM and cerebral WM volumes also correlated negatively with walking times; and volumes of thalamus, subcortical GM, and cortical GM correlated with both average and maximum hand grip strength. Total subcortical GM volume is plotted against maximum grip strength in Fig. (2). Correlations of regional brain volumes with hand grip strength tended to be slightly stronger when maximum rather than average grip strength was used. The caudate nucleus, putamen, pallidum, hippocampus, amygdala and nucleus accumbens showed no volumetric correlations with any of the three continuous frailty variables.
| Brain region | Average grip strength | Maximum grip strength | Walking time |
|---|---|---|---|
| Thalamus | 0.51 (0.002) | 0.51 (0.002) | ns |
| Cortical GM | 0.45 (0.007) | 0.48 (0.004) | ns |
| Subcortical GM | 0.49 (0.003) | 0.52 (0.001) | −0.40 (0.020) |
| Cerebellar WM | ns | ns | −0.63 (<0.001) |
| Cerebellar GM | 0.40 (0.016) | 0.44 (0.008) | −0.39(0.023) |
| Cerebral WM | 0.43 (0.011) | 0.46 (0.005) | −0.35 (0.044) |
Controlling for age, ICV, and nadir CD4 count or BDI score attenuated these relationships as assessed by predictive regression analyses in which regional volumes were the outcomes. (Age correlated significantly with volumes of the thalamus, caudate, pallidum, amygdala and subcortical GM [R~ −0.4], and at trend-level for the putamen, hippocampus, nucleus accumbens, cortical GM, cerebral WM, cerebellar GM and cerebellar WM [R~ −0.3]). Regional brain volumes were regressed against walking time, average grip strength and maximum hand grip strength (separately). (The time since HIV diagnosis was tested as a covariate, but had no significant effect and was not retained.) Increased walking time (slower gait) was associated with reduced cerebellar WM (p<0.001, β= −0.60) and total subcortical GM (p=0.036, β= −0.30) volumes independently of age, ICV and nadir CD4 count (Table 3). Reduced thalamus volume was independently related to weaker maximum (p=0.015, β=0.42) and average (p=0.022, β=0.39) hand grip strength. Similarly, caudate volume was negatively associated with maximum (p=0.007, β= −0.48) and average (p=0.008, β= −0.46) grip strength values.
The effects of depressive symptoms were investigated by replacing nadir CD4 by BDI-TOT as a covariate. Relationships of average and maximum hand grip strength with regional brain volumes were virtually unchanged, although associations with thalamus volume became slightly weaker and less significant (results not shown). Use of BDI-S or BDI-CA in place of BDI-TOT made little difference. Because of collinearity with walking time, BDI scores were not tested as covariates when walking time was an independent variable. The associations in Table 3 were less significant if adjusted for age and ICV only (i.e., if neither nadir CD4 nor BDI-TOT was a covariate). Additionally, we note that when the regression models controlled for both nadir CD4 and BDI-TOT in addition to age and ICV, significant brain volumetric associations with hand grip strength were unchanged (for thalamus) or became more significant (for caudate), and no non-significant relationships became significant. These results are not shown in Table 3 and must viewed with caution because of possible sample size issues.
| Dependent variable | Average grip strength | Maximum grip strength | Walking time | ||||||
|---|---|---|---|---|---|---|---|---|---|
| p-value | β | R2 | p-value | β | R2 | p-value | β | R2 | |
| Caudate† | 0.008 | −0.46 | 0.42 | 0.007 | −0.48 | 0.43 | ns | - | - |
| Thalamus† | 0.022 | 0.39 | 0.42 | 0.015 | 0.42 | 0.44 | ns | - | - |
| Subcortical GM | ns | - | - | ns | - | - | 0.036 | −0.30 | 0.51 |
| Cerebellar WM | ns | - | - | ns | - | - | <0.001 | −0.60 | 0.41 |
Table 4 presents the results of explanatory regression modeling that evaluated effects of regional brain volumes (independent variables) on continuous frailty parameters, adjusting for age. Because of its collinearity with regional volumes, we did not use ICV as a covariate. BDI scores were tested as independent variables. When grip strength values were the outcome variables, BDI scores had no effect (as expected, because Pearson correlations of BDI-TOT, BDI-S and BDI-CA with hand grip strength were non-significant) and therefore were not included in analyses. Volumes of thalamus, subcortical GM, cortical GM, cerebral WM and cerebellar GM were associated with average and maximum hand grip strength (Table 4a). The relationships, unaffected by depressive symptoms, were similar to the corresponding univariate Pearson correlation values despite adjustment for age. Adjusted R2 values for the models were low, perhaps because each of the five regional volumes was considered in a separate regression analysis.
Total and component BDI scores were retained as covariates when walking time was regressed against regional brain volumes and subject age (Table 4b). Inclusion of BDI-TOT gave the best overall results (adjusted R2 of the models). Decreased WM volumes related to slow gait independently of age and depressive symptoms. Reduced cerebellar WM volume was associated with longer walking time regardless of whether BDI-TOT, BDI-CA or BDI-S was used (p<0.001, β~ −0.6). The adjusted R2 values suggest that cerebellar WM atrophy may play a causal role in slow gait speed. Cerebral WM volume was significantly related to walking time independently of age and BDI-TOT or BDI-S, but the association became a trend upon adjustment for the BDI cognitive/affective component (p=0.07, β ~ −0.3).
DISCUSSION
In chronically HIV-infected older adults on stable cART, reduced walking speed and hand grip strength were linked to decreased volumes of brain regions that control motor and cognitive function. These atrophic changes may signify the early pathology associated with development of frailty-related motor dysfunction, given that frailty was less prevalent in our subjects than reported in other HIV patients. Few studies have investigated relationships between brain structure and frailty measures in HIV, although one group observed correlations between MRI-detectable brain atrophy and hand grip strength (measured during neuropsychological testing rather than to gauge frailty) [18]. We examined such associations by multivariate regression analyses that used continuous frailty parameters as independent variables and regional brain volumes as outcomes, and vice versa.
Our most robust result was the association between gait speed and cerebellar WM volume. With the caveat that the data were cross-sectional, longer walking times predicted decreased cerebellar WM volumes independently of age, ICV and nadir CD4. Slower gait speed was similarly predictive of reduced subcortical GM volume. Conversely, causal inference based on explanatory modeling indicated that cerebellar WM atrophy may lead to reduced gait speed independently of depressive symptoms and age. Cerebral WM volume also appeared to play a role in slow gait but had less explanatory power as determined by the model’s adjusted R2. These findings are in broad agreement with links reported between brain volumetric loss and slow gait in large geriatric cohorts. Among 2450 high-functioning adults in the longitudinal Cardiovascular Health Study (CHS), severe brain ventricular enlargement indicative of global atrophy was associated with greater slowing of gait and incident functional impairment over 5 years [32].
Slow gait has emerged as a major predictor of disability and morbidity [33-35]. Information processing, which is central to multiple cognitive processes [36], is closely linked to gait speed [37, 38]. The Mayo Clinic Study of Aging analyzed 1478 cognitively normal elderly participants and found faster gait to be cross-sectionally associated with better performance in memory, executive function, and global cognition [29]. Although both gait speed and neuropsychological test scores decreased over time, faster gait at baseline was longitudinally associated with less cognitive decline globally and across all NP domains [29]. Importantly, baseline cognition did not relate to altered gait speed, implying that slowed gait precedes cognitive impairment [29].
Gait slowing has been associated with poorer learning and memory [39] and may be a sensitive indicator of subclinical cognitive deficits [29]. Global cognitive function in our study population was relatively unimpaired. There were no significant NP differences between frail-risk and non-frail subjects, perhaps due to the small size of the former group. However, frail-risk patients showed a reduction in learning and memory performance that approached significance when only the pre-frail individuals were considered. Both frail-risk and pre-frail subjects demonstrated significantly weaker hand grip strength. Interestingly, hand grip strength was associated (in predictive models) with volumes of thalamus and caudate, brain regions that are implicated in learning and memory [40, 41]. Learning and other higher cognitive processes, as well as motor control, are also subserved by the cerebellum [42, 43]. Larger cerebellar GM volume predicts general cognitive ability [44] and correlates with faster gait and information processing [45] in older adults. Cerebellar GM atrophy is associated with executive dysfunction [46]. Unlike the HIV-negative individuals in those studies, however, our HIV+ subjects demonstrated stronger correlations of gait speed with cerebellar WM volume than with cerebellar GM. Cerebellar GM reduction was recently proposed as a signature of physical frailty in HIV-uninfected community-dwellers aged 50 years and older [47]. Our data, in contrast, suggest that cerebellar WM decrease should be examined as a correlate of frailty in chronic HIV infection. Furthermore, cerebellar WM pathways are known to be involved in processes such as verbal working memory [48] and the rate of learning visuomotor tasks [49]. Although physical frailty has been related to cognitive decline cross-sectionally [50] and longitudinally [51, 52], additional work is needed to clarify how the development of frailty may be associated with declines in learning and memory in the setting of chronic HIV.
Since the caudate nucleus controls the initiation and execution of somatic motor activity (especially willed movement) [53], this structure might be expected to show atrophy related to lower hand grip strength. Instead, we found that larger caudate volumes were associated with weaker grip. This association may well be an artifact of low sample size. Alternatively, because our patients had well-controlled HIV infection and only mild cognitive impairment, the result may indicate initial hypertrophy of the caudate nucleus which will likely change to atrophy as the disease progresses. A similar phenomenon is seen in individuals with presymptomatic familial Alzheimer’s disease, who exhibit caudate hypertrophy that reflects inflammation and precedes atrophy [54]. In HIV, enlarged basal ganglia volume has been associated with mild cognitive impairment and motor slowing [55]. The relationship that we identified between weaker hand grip and larger caudate size may be a manifestation of the hypermetabolism that occurs (as a possible compensatory response) in the basal ganglia of HIV patients who have subclinical motor deficits or mild cognitive impairment [56, 57]. In chronic HIV disease, caudate enlargement and hypermetabolism may play a role in frailty-related cognitive decline.
As noted above, decreased cerebral and cerebellar WM volumes appeared to be causative factors in slow gait speed. Reduced volumes of thalamus, GM (cortical, cerebellar, total subcortical) and cerebral WM may contribute to loss of hand grip strength, but the R2 values for these explanatory models were low, probably because of omitted variables and because regional volumes were evaluated in separate analyses. Studies with large N must resolve these issues and determine the etiology of weakened hand grip strength and other frailty characteristics in HIV patients.
Cross-sectional studies of frailty and pre-frailty in HIV-uninfected, community-dwelling older individuals (reviewed by Collard et al. [58]) report disparate prevalence estimates. Those studies involved participants aged 65 and older [58], and while their frailty rates were higher than ours (4 – 17% vs. 3%), their pre-frailty prevalence (18.7% – 47.9%) was in line with what we observed (23%). Because frailty in HIV disease is associated with older age [12], the patients in our study, on average 51 years old, may be developing the condition; some who are currently in the pre-frail category may become frail by age 65. Rates of frailty and pre-frailty were greater in the larger HAHC-CVD cohort (our parent study) than in the sub-study. Of the 69 HAHC-CVD participants with frailty data, 9% were frail and 26% were pre-frail (unpublished data). Subject ages did not differ between the sub-study and parent study. Many of the frailer HAHC-CVD participants declined to undergo MRI for the sub-study.
Frailty studies in HIV-uninfected individuals under 65 years of age are scarce. Estimates of frailty and pre-frailty rates among younger individuals (≥ 45 years old) were recently published by Kooij et al. [59]. The Fried frailty phenotype was assessed in 521 HIV-infected individuals (of whom 93% were virally suppressed) and 513 HIV-uninfected controls in the AGEhIV Cohort Study [59]. HIV infection was independently associated with pre-frailty and frailty: among the HIV-negative participants (median age of 52 years), ~3% were frail and 36% pre-frail, compared with ~11% and51% of HIV+ individuals who were frail and pre-frail, respectively. Relative to the HIV+ participants of the AGEhIV Cohort Study, frailty and pre-frailty prevalence in our HIV patients is considerably lower; i.e., similar to that of the HIV-negative AGEhIV controls. Our results may be skewed by small sample size. The occurrence of frailty during middle age should be further investigated in healthy as well as in patient populations.
Data more comparable to ours were reported by a large (N=445) cross-sectional study of HIV-infected urban outpatients (mean age=42 years; 75% on cART) who were clinically and demographically similar to our subjects [12]. Frailty prevalence was 9%, and frail and non-frail groups did not differ in gender, use of cART, or viral suppression (undetectable viral load) [12]. The lower frailty prevalence that we found may reflect our smaller sample size, the higher proportion of males in our study (88.5% vs. 70.8%), and the fact that 100% of our participants were on cART and more (77% vs. 69%) were virally suppressed. Women in the general population have higher (worse) frailty scores compared to men of the same age [60]. Among HIV patients, frailty has been associated with detectable viral load [61]. Long-term cART reduces the likelihood of developing frailty [62]; shorter duration of therapy is linked to higher frailty prevalence [11]; and longer treatment is protective even after controlling for CD4 count [62].
Frailty in cART-treated HIV patients has also been related to co-morbidity and past opportunistic illnesses [12], low bone and muscle mass [63], and other factors (e.g., longer in-patient hospital stays and lower socioeconomic status) [12] not considered in our analysis. Co-morbid conditions associated with frailty in this population include chronic viral hepatitis, chronic renal failure, cardiovascular disease, and neuropsychiatric disorders [12]. Frail HIV patients exhibit reductions in serum albumin that may be a manifestation of chronic low-grade inflammation [12].
Further research is required to determine rates of frailty in the era of cART, in part because of differing assessment tools and conflicting data. Without a standard definition of frailty in HIV disease, the most commonly used scale is the frailty-related phenotype of Fried and colleagues [23], often modified. In the Multicenter AIDS Cohort Study (MACS), while initiation of cART was associated with decreased frailty when the assessment excluded grip strength [11], prevalence estimates among cART-treated patients rose following the addition of grip strength to the MACS frailty scale [61]. For development of an HIV-specific frailty index, consideration should be given to potentially useful measures that detect existing or incipient frailty. Gait patterns (step-to-step variability of gait parameters) may be more indicative than slow gait of early mobility impairment. Extreme step width variability correlates with a history of falling in older, HIV-uninfected individuals who have normal gait speed [64], and high stride time variability predicts falls [65]. The high-functioning elderly exhibit subclinical brain vascular abnormalities that are associated with increased variability of step length in an apparent compensatory mechanism regulated by the basal ganglia [66]. Frailty markers other than average hand grip strength and average gait speed may be appropriate for assessing HIV patients.
The one subject categorized in our study as frail was male and virally suppressed (factors associated with a decreased risk of frailty), but had a strikingly low nadir CD4 count of 8.0 cells/mm3, consistent with the reported link between low nadir CD4 and frailty in cART-treated HIV patients [12]. The frail subject’s current CD4 count was 273.0 cells/mm3, supporting the claim that frailty in HIV is strongly associated with low CD4 [62, 67]. His total BDI score of 14 indicated mild depression. The 8 pre-frail subjects were all virally suppressed and did not differ significantly from the non-frail group in demographics, regional brain volumes or clinical variables (including current and nadir CD4 count). Although this may be a consequence of low sample size, it suggests that traditional HIV-related factors are not the most useful in identifying development of physical frailty or patients who are vulnerable.
Obvious limitations of the present study are its cross-sectional design, small sample size, and lack of an HIV-uninfected comparison group. Larger studies may reveal regional brain volumetric group differences between non-frail and frail (or even pre-frail) individuals. Longitudinal observation of large cohorts is necessary to model the emergence of frail and pre-frail conditions. Changes that may have occurred between the frailty assessment and brain MRI, which were performed 1 year apart on average, were potential confounding factors. (In the CHS, however, adjustment for the approximately 9-month interval between MRI and gait examination did not alter relationships between regional GM volumes and gait measures [68].) Among our study’s strengths was the consideration of depression (with effects of cognitive/affective and somatic aspects examined separately) as well as of age and nadir CD4. Larger studies are warranted as little is known about the development or progression of frailty in HIV-infected persons on suppressive cART; frailty risk factors among aging patients with undetectable viral load are uncertain [69]. Recent work shows frailty to be transient or reversible, especially in younger HIV patients who can revert to their pre-frail state [62, 67]. Frailty may be ameliorated by improving CD4 count, and early treatment is critical [62, 67]. Moreover, as indicated by our study, characteristics of frailty are linked to structural changes in the brain. Gait speed is a simple, inexpensive performance measure that may serve to identify HIV patients at risk for brain atrophy or HAND.
CONCLUSION
To summarize, we identified cross-sectional associations of regional brain volumes with walking speed and hand grip strength. Volumetric changes in cerebellar white matter and subcortical gray matter (especially thalamus and caudate), may be connected to the development of pre-frail and frail conditions in HIV patients on suppressive cART. Gait speed, especially, is sensitive to alterations in white matter, and slow gait should be investigated as an early predictor of brain atrophy and physical frailty. If validated in larger studies, our results may improve understanding of the mechanism underlying premature frailty in well-controlled HIV disease, ultimately facilitating prevention or management of cognitive difficulties and the frailty syndrome.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
We thank our study participants for their contributions and the staff at InVision Imaging for support and assistance. This study was supported by the National Institutes of Health (R01HL095135 [C. Shikuma] and U54MD007584 [J. Hedges]).


