All published articles of this journal are available on ScienceDirect.
Changes in Serum High-Molecular-Weight Adiponectin Levels in Critically Ill Children with Systemic Inflammatory Response Syndrome
Abstract
Background:
The aim of the study was to evaluate the effect of the systemic inflammatory response syndrome (SIRS) on serum high-molecular-weight adiponectin (HMWA) levels.
Method:
Twelve children with SIRS were enrolled in this study at the intensive care unit (ICU). Twelve age and sex matched healthy subjects were selected as controls. Serum HMWA, interleukin-6 (IL-6), C-reactive protein (CRP), and procalcitonin (PCT) levels were determined after 2 hours and 24 hours, and on the day of discharge which was on the average 9.4 days after ICU admission.
Results:
2 hours after admission to the ICU, the patients had significantly decreased serum HMWA levels compared with healthy controls (P < .001). 24 hours after the admission, the patients did not have any significant changes in their HMWA levels, however on the day of discharge, on average 9.4 days after hospital admission, a significant increase was observed (P < .05). After the treatment, there was a decrease in serum PCT, IL-6 and CRP levels. The only variable that was decreased 24 hours after the ICU admission was PCT (P < .05). A negative correlation was found between serum HMWA and PCT levels, and between HMWA and CRP (P < .05 and P < .01), however no correlation was found between HMWA and IL-6.
Conclusion:
In SIRS we observed a marked reduction in serum HMWA concentrations and a profound increase in IL-6, PCT, and CRP levels. A significant relationship between serum HMWA and PCR and CRP levels was evident.
INTRODUCTION
The adipocyte-specific protein adiponectin circulates in high concentrations in human plasma [1]. It has anti-inflammatory, antioxidant, antiatherosclerotic and vasoprotective effects and might be involved in the pathogenesis of the systemic inflammatory response during sepsis [2]. Adiponectin - the prototype of an anti-inflammatory adipokines and the most abundant adipokine is diminished in sepsis [3]. Low levels of adiponectin have been found in critically ill patients, although data are limited in human subjects at this stage. The role of adiponectin in systemic inflammation and critical illness is not well defined. Early data suggest that plasma levels of adiponectin are decreased in critical illness. Whether this is a result of the disease process itself, or whether patients with lower levels of this hormone are more susceptible to developing a critical illness is not known [1]. Serum high-molecular-weight adiponectin (HMWA), the biologically most relevant molecule, has been investigated very little in human sepsis [4]. The aim of the study was to evaluate the effect of systemic inflammatory response syndrome (SIRS) on HMWA levels.
MATERIALS AND METHODS
Participants
A prospective study was conducted. Twelve children (average age 9.4 years (13 - 209 months); 4 female and 8 male) with SIRS (including 7 patients with sepsis, but without septic shock) were enrolled in this study at the intensive care unit (ICU) of Children Clinical University Hospital, Riga, Latvia. Children had SIRS according to the International consensus conference on pediatric sepsis definition – at least two of below mentioned criteria, one of which must be abnormal temperature or leukocyte count: 1). Core temperature >38.5 or <36.0°C; 2). Tachycardia, defined as a mean heart rate >2 SD above normal for age; or for children <1yr old, bradycardia, defined as a mean heart rate <10th percentile for age; 3). Mean respiratory rate >2 SD above normal for age; 4). Leukocyte count elevated or depressed for age or >10% immature neutrophils [5]. Patients were admitted on average 3, 5th day of illness (minimal value 1, maximal - 14 days). All patients received intravenous fluid therapy, which was initiated upon arrival in the ICU, 10 out of 12 children received antibacterial therapy, which was initiated on median the 1st day after admission (minimal value 1st day, maximal - 4th day, average 1,41). 9 patients had confirmed infection (by radiological or microbiological examination). Twelve for age and sex matched healthy subjects were selected as controls.
Patient’s exclusion criteria from the study were antibacterial therapy within the last 48 h, immunodeficiency, chronic liver or kidney illness, vaccination within 5 days before the start of the illness, congenital metabolic defects, chromosomal anomalies, and use of corticosteroids or immunosuppressant medications. Patients with obesity, diabetes mellitus and other diseases which are known to be associated with significant changes of anti and pro-inflammatory biomarkers, including surgery or trauma within the preceding 30 days, were not included.
All subjects gave their informed consent to the protocol, which was approved by the local Medical Ethics Committee of the Riga Stradins University, Latvia.
Analysis
The serum HMWA was determined with enzyme-linked immunosorbent assay (Infinite F200 pro, TECAN, Salzburg, Austria), interleukin-6 (IL-6) levels were determined with a chemiluminescent immunometric assay (Immulite 2000, Siemens Medical, Germany), C-reactive protein (CRP) level was determined by the Latex method (COBAS INTEGRA; Roche professional Diagnostics) and procalcitonin (PCT) level was determined by enzyme-linked fluorescent assay (VIDAS B.R.A.H.M.S, bioMérieux, France) - 2 hours, 24 hours, and on discharge average 9.4 days after ICU admission.
After testing the normality of data distribution with the Kolmogorov-Smirnov test (HMWA and all other biomarkers had a normal distribution), statistical differences between two groups were analyzed by the two-sided unpaired Student's t test, and differences between three groups assessed by Friedman ANOVA and Kendall's Coefficient of Concordance. Data were recorded as the mean±SD and two-tailed values of p=0.05 was considered to be significant. Correlation analyses were performed using one-factor linear regression analysis. All analyses were performed using STATISTICA 10 software (StatSoft Inc., USA).
RESULTS
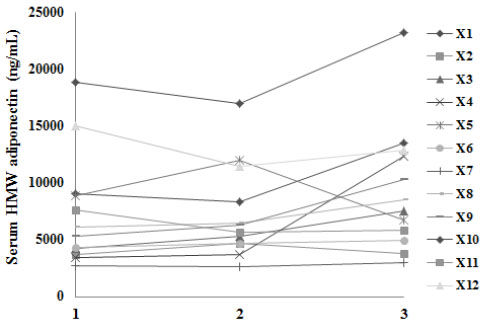
2 hours after admission to the ICU, the patients had significantly decreased serum HMWA levels compared with healthy controls (7452.6±4958.91 vs. 20236.42±2817.47 ng/ml; p<0.001).
24 hours after the admission, the patients did not have any significant changes in their HMWA levels, however there was a significant increase in HMWA on the day of discharge on average 9.4 days after hospital admission (p<0.05) (Fig. 1).
Serum levels of PCT, IL-6 and CRP were also decreased as a result of treatment (Table 1), however, the only variable that was decreased 24 hours after ICU admission was PCT.
There were negative correlations between serum HMWA and PCT, and between HMWA and CRP, but there was no correlation between HMWA and IL-6 (Table 2).
DISCUSSION
The data of our preliminary study show that serum HMWA levels are decreased in SIRS patients (with and without sepsis), this is in concordance with the data of other studies [4, 6-8], which have also provided evidence that the adiponectin level is significantly lower in patients with higher pediatric risk of mortality score and that plasma adipokines correlate with the risk of mortality scores in sepsis and therefore may be useful biomarkers [6]. The findings suggest a protective role of adiponectin in diminishing microvascular organ-specific endothelial cell activation during sepsis [9].
| Biomarker | 2 hours after ICU admission | 24 hours after ICU admission | On the day of discharge, on average 9.4 days after ICU admission | p-value* |
|---|---|---|---|---|
| PCT (ng/ml) | 11.01 ± 11.63 | 7.01 ± 7.30** | 3.55 ± 6.37 | 0.011 |
| IL-6 (pg/ml) | 183.36 ± 293.98 | 50.49 ± 59.95 | 9.50 ± 9.64 | 0.009 |
| CRP (mg/l) | 153.10 ± 141.62 | 110.96 ± 81.91 | 54.32 ± 96.03 | 0.046 |
* p < 0.05 compared to all group. ** p = 0.04 vs 2 hours after ICU admission.
| HMWA | PCT | IL-6 | CRP | |
| HMWA | — | |||
| PCT | r = -0.34; p = 0.047 |
— | ||
| IL-6 | NS | r = 0.46; p = 0.006 |
— | |
| CRP | r = -0.46; p = 0.004 |
r = 0.83; p = 0.00000 |
r = 0.54; p = 0.0008 |
— |
It appears that an inverse relationship exists between adiponectin and inflammatory biomarkers [1], although the results of other study have shown that inflammatory cytokines are not correlated with serum adiponectin [10]. Our results also correspond with the findings that HMWA is negatively correlated with PCT [11] but not with IL-6 [12].
HMWA play a role in diagnosis of infection and predicting the outcomes, and is correlated with the severity of sepsis [11]. Observations of lower adiponectin levels, then raises the possibility of therapeutic options to increase circulating adiponectin levels [1].
A critical role of adiponectin in sepsis-related inflammation is likely through a multifactorial anti-inflammatory mechanism that serves to limit leukocyte infiltration, chemotaxis, and endothelial activation. Evidence suggests that clinical conditions associated with hypoadiponectinemia, may predispose to sepsis-related complications through altered adipokine homeostasis [13]. Interestingly, low adiponectin levels at the medical ICU admission were an independent positive predictor of short-term and overall survival [10].
Presumably, adipose tissue and its products may have some impact on numerous pathways of response to trauma, sepsis and stress. The discussion on a plausible role of adiponectin in critical illness has been raised by the fact of finding hypoadiponectinemia in critically ill patients. The nature of this phenomenon, however, remains to be elucidated, and noteworthy clinical studies should prompt further efforts in basic research to explain the mechanisms beyond the clinical observation of low adiponectin levels in humans with severe illness [14].
The simultaneous secretion and release of multiple mediators such as pro-inflammatory biomarkers (and simultaneously decreased release of anti-inflammatory cytokines) were found to be involved in the initiation of systemic inflammation in sepsis. Therefore, measuring the concentration of multiple mediators may help in the early detection of sepsis and help to avoid unnecessary antibiotic treatment [15].
CONCLUSION
In conclusion, the study has proved that it is possible to determine a SIRS (with/without sepsis) specific adipocytokine profile (HMWA) in critically ill patients. In SIRS and, possible sepsis patients, we observed a marked reduction in serum HMWA concentration and a profound increase in IL-6, PCT, and CRP levels. A significant relationship between serum HMWA and PCR and CRP levels was evident. These data raise the possibility that HMWA may play a part in the inflammatory response in patients with severe illness.
LIST OF ABBREVIATIONS
| CRP | = C-reactive protein |
| HMWA | = High-molecular-weight adiponectin |
| ICU | = Intensive care unit |
| IL-6 | = Interleukin-6 |
| PCT | = Procalcitonin |
| SIRS | = Systemic inflammatory response syndrome |
CONFLICT OF INTEREST
The authors declare that there are no conflicts of interest regarding the publication of this manuscript. Funding of the research played no role in the study design; collection, analysis, and interpretation of data; preparation of this manuscript; or in the decision to submit the report for publication.
ACKNOWLEDGEMENTS
Funding by the grant No. 2014.10-4/VPP-5/16 BIOMEDICINE of the framework of the Latvian National Program.


