All published articles of this journal are available on ScienceDirect.
Dosimetrical Correlation between Cervical Site Radiation Dose and Radiation Dermatitis in Advanced Nasopharyngeal Carcinoma: A Retrospective Analysis
Abstract
Introduction
To investigate the dosimetric correlation between radiation dose to the cervical site and radiation-induced dermatitis in patients with advanced nasopharyngeal carcinoma undergoing concurrent chemoradiotherapy.
Methods
Data from 109 patients treated with concurrent chemoradiotherapy between November 2018 and June 2020 were analyzed. The study focuses on identifying clinical and dosimetric factors associated with moderate to severe radiation dermatitis and determining the optimal radiation dose to the cervical site.
Results
Univariate analysis revealed that nodal stage, tumor node metastasis stage, volume to gross tumor volume of the neck, and mean dose to the cervical site were associated with an increased risk of grade 3/4 radiation dermatitis. Multivariate analysis identified cervical site of mean dose (odds ratio=1.003; 95%confidence Interval=1.001-1.005; P=0.003) as an independent predictor. The optimal cut-off value of cervical site as was 5206 centigray (area under curve =0.733%; P<0.01;95% confidence Interval =0.637-0.829).
Discussions
While prior studies have established an association between cervical radiation dose and dermatitis severity, detailed radiotherapy dose-specific data regarding radiation dermatitis remain scarce. This study aims to clarify in detail the toxicity levels of such skin damage across different radiation doses and provides precise dosimetric insights.
Conclusion
Moderate or severe radiation dermatitis is significantly correlated with the mean dose to the cervical site, and keeping the cervical dose below 5200 centigray may decrease the incidence of severe skin toxicity. We recommend patients with mean dose over 5200 centigray to consider topical treatment during the early stages of radiotherapy and advanced techniques to reduce radiation volumes and doses to the cervical site may be useful.
1. INTRODUCTION
Nasopharyngeal carcinoma (NPC), originating from the mucous membrane of the nasopharynx, exhibits the highest incidence in southern China. Due to its high sensitivity to ionizing radiation, radiotherapy is the preferred treatment modality [1]. Advances in radiotherapy technologies have significantly reduced the mortality of NPC, with reported 5-year relapse-free survival (RFFS) rates of 90.2% to 98.5% and overall survival (OS) rates of 76.4% to 91.7% [2]. Studies indicate 5-year overall survival rates ranging from 70.5% to 89.4% for patients with stage II to IVB disease [3-5]. Long-term survival among NPC patients underscores the importance of enhancing quality of life, as survivors may face complications such as cervical subcutaneous fibrosis, hearing loss, and skin dystrophy [6, 7].
In this study population, NPC incidence was relatively low, yet most cases were diagnosed at stage III or higher, posing challenges in radiotherapy planning. Addressing these challenges could lead to significant clinical advancements. We therefore investigated adverse events associated with chemoradiotherapy, explored correlations between clinical parameters and radiation dermatitis, and specifically investigated the dosimetric relationship between radiation doses to the neck site and radiation dermatitis in NPC patients. Potential strategies for managing radiation dermatitis are also discussed.
2. MATERIALS AND METHODS
This retrospective case-control study analysis included 109 patients diagnosed with NPC (stage II-IVA, AJCC 8th edition) treated at Anhui Provincial Cancer Hospital from November 2018 to June 2020. The cohort comprised 71 males and 38 females, aged 21 to 75 years, with a median age of 50.4 years. Patient characteristics are presented in Table 1. Eligibility criteria were as follows:
| All Patients (N=109) | Mild | Moderate/Severe | P | |
|---|---|---|---|---|
| Tumor Stage Group (AJCC8th) | - | - | - | - |
| II | 16(14.68%) | 10 | 6 | 0.046* |
| III | 62(56.88%) | 27 | 35 | - |
| IVa | 31(28.44%) | 8 | 23 | - |
| Tumor(T) | - | - | - | - |
| T1 | 6(5.50%) | 1 | 5 | 0.482 |
| T2 | 48(44.04%) | 23 | 25 | - |
| T3 | 46 (42.20%) | 18 | 28 | - |
| T4 | 9 (8.26%) | 3 | 6 | - |
| Lymph node | - | - | - | - |
| N1 | 36(33.03%) | 19 | 17 | 0.046* |
| N2 | 49(44.95%) | 21 | 28 | - |
| N3 | 24 (22.02%) | 5 | 19 | - |
| Pathological pattern | - | - | - | - |
| Nonkeratinizing squamous cell carcinoma | 82(75.23%) | 37 | 45 | 0.596 |
| Squamous carcinoma | 18(16.51%) | 6 | 12 | - |
| Poorly differentitated carcinoma | 9(8.26%) | 3 | 6 | - |
| Gender | - | - | - | - |
| Male | 71(65.14%) | 30 | 41 | 0.779 |
| Female | 38(34.86%) | 15 | 23 | - |
| Age(y) | - | - | - | - |
| <30 | 4(3.67%) | 2 | 2 | 0.932 |
| 30-59 | 84(77.06%) | 35 | 49 | - |
| ≥60 | 21(19.27%) | 8 | 13 | - |
Note: (AJCC8th) =American joint committee on cancer.
(1) Karnofsky Performance Status (KPS) ≥ 70; (2) sufficient organ function and capacity for regular follow-up; (3) biopsy-confirmed NPC (via endoscopic biopsy or cervical lymph node biopsy); and (4) initial evaluation included MRI/CT scans of the nasopharynx and neck, chest CT, upper abdominal ultrasound, bone ECT scan, and laboratory tests (complete blood count, biochemical profile, and EBV DNA). Exclusion criteria were as follows:
(1) Karnofsky Performance Status (KPS) <70; (2) severe or uncontrolled concurrent medical conditions; (3) pregnancy at diagnosis; (4) prior history of head and neck radiotherapy.
Sample size was calculated using G*Power 3.1 software with the following parameters: effect size = 0.3, α = 0.05, and statistical power (1−β) = 0.8. Incorporating the disease incidence in the study area (where the study population resided), the total required sample size was determined to be no less than 102 cases. Clinical data of the cases were extracted from hospital electronic medical records, and the eligibility criteria for this study were established in accordance with previously published literature [9] [8-10].
2.1. Chemotherapy Regimens
Induction chemotherapy consisted of taxel (or docetaxel) in combination with cisplatin (or nedaplatin). All patients received 1-3 cycles of chemotherapy regimens every 3 weeks (depending on the patient's physical condition and tolerance).
2.2. Concurrent Chemoradiotherapy
Concurrent chemoradiotherapy included cisplatin (or nedaplatin)-based regimens, with intravenous cisplatin (or nedaplatin) at 40 mg/m2 weekly, plus intensity-modulated radiotherapy (IMRT) based on individual performance status and physician discretion. Among 109 patients, 106 received cisplatin, and 3 received nedaplatin over 3-5 cycles of chemotherapy.
2.3. Radiotherapy Features
The planning computed tomography (CT) dataset was acquired using a 16-detector scanner (GE Healthcare, Chicago, IL, USA). CT image data were reconstructed as 2.5-mm slices for IMRT and VMAT (Varian Medical Systems, PaloAlto, CA, USA). Treatment with radiotherapy technologies and fractionated dose with an individually optimised plan for each patient was mainly considered according to the NCCN (National Comprehensive Cancer Network) guidelines, with gross tumor volume (GTV), gross primary tumor (GTVnx), and involved lymph nodes (GTVnd) defined according to the magnetic resonance imaging (MRI) or contrast-enhanced CT, clinical, and endoscopic examinations.
For all patients, the latest MRI, or CT images were adopted to delineate the gross tumor volume (GTV). GTVnx was defined as the primary tumor site and posterior pharyngeal lymph nodes. GTVnd was defined as cervical lymph nodes identified on imaging, regardless of whether they were positive or negative. The GTVnx, plus a 3 mm margin, was defined as planned gross tumor volume (PGTVnx), and the GTVnd, plus a 3 mm margin, was defined as PGTVnd. Radiation oncologists categorised CTV (clinical target volume) into CTV1 (high risk) and CTV2 (low risk). CTV1, defined as high-risk areas, should include the entire nasopharynx, retropharyngeal lymph nodal regions, and any high-risk nodal regions. CTV2, defined as low-risk areas, was recognized as the next station of the positive lymph nodes in the selected neck area. The other two planning target volumes (PTV1 and PTV2) were defined as CTV1 plus a 3 mm margin and CTV2 plus a 3 mm margin, respectively. The total radiation dose (delivered in 28-33 fractions) was 60-70 Gy to the primary tumor, 61-70 Gy to the cervical lymph nodes, 54-63 Gy to the high-risk region, and 50-56 Gy to the low-risk and neck node regions.
All radiotherapy regimens have delineated and jointly reviewed by two certified radiation oncology physicians. Target volume and dose variations, and dose-volume histogram statistics for all patients (extracted from Varian Radiotherapy Treatment Planning System). For each patient, the cervical site was outlined separately from the skull base to the thoracic inlet on planning CT images by a radiation oncologist. Mean dose (Dmean) and dose-volume histograms (DVH) with 1 Gy bins were analyzed to assess the association between severe RD and Dmean of the cervical site.
2.4. Radiation Dermatitis Assessment
Assessments should be conducted by two radiation oncologists experienced in managing radiation-related skin toxicities to ensure consistent interpretation of skin changes. Evaluate the cervical skin wound conditions immediately after completion of radiotherapy and then weekly until treatment ends. Radiation dermatitis is graded using the Radiation Therapy Oncology Group (RTOG) criteria (Table 2), which categorizes cases into grades 0–4. Grades 1–2 are defined as “mild,” and grades 3–4 as “moderate/severe.”
| 0 | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| No charge | Faint erythema | Tender bright erythema | Moist desquamation in areas other than skin folds | Olceration | Death |
| - | Dry desquamation | Moderate edema patchy moist | Pitting edema | Hemorrhage | - |
| - | Epilation | Desquamation | - | Necrosis | - |
| - | Decreased sweating | - | - | - | - |
Note: RTOG= Radiation therapy oncology group.
3. OUTCOME
3.1. Treatment Efficacy
Treatment efficacy was assessed based on local control rates, including complete remission (CR), partial remission (PR), and stable disease (SD). Overall survival (OS, calculated as the time from the start of treatment until death from any cause). The secondary endpoints were distant metastasis-free survival (DMFS), locoregional relapse-free survival (LRFS), and disease-free survival (DFS).
3.2. Follow-up
All patients were followed up regularly every 3–6 months from the end of radiotherapy until December 31, 2023. All 109 patients (100%) completed the planned follow-up. Physical examinations, nasal endoscopy, imaging studies (chest, abdominal ultrasound, nasopharyngeal MRI, and bone scan) were done in each follow-up.
3.3. Statistical Analysis
Clinical characteristics (age, gender, pathological pattern, AJCC stage) and dosimetric features (Dmean to cervical site, Dmean doses and volumes of GTV and GTV-nd, shown in (Tables 1 and 3) were analyzed. Kaplan-Meier curves estimating PFS, OS, DMFS, and LRFS were established. Table 4 shows the doses and volumes prescribed for PGTV, PGTV-nd, PTV1, and PTV2 for all participants. All patients received radiotherapy on both sides of the neck. Statistical analyses were done using SPSS version 26.0, with comparisons of moderate/severe radiation dermatitis incidence across different mean cervical doses using the Chi-square or Fisher's exact test. Variables with P < 0.1 in univariate analysis were included in a multivariable model. Binary logistic regression was done to assess the relationship between moderate/severe RD and different variables. Areas under the curve (AUC) for dosimetric parameters predicting grade > 2 (moderate or severe) RD were calculated by Receiver operating characteristic (ROC) curve analysis. Statistical significance was set at P < 0.05 (two-tailed).
| Stage | Mild Average(min~max) | Moderate/Severe Average(min~max) |
P |
|---|---|---|---|
| II | - | - | - |
| Dmean to cervical | 51.69(49.81~54.66) | 54.73(52.43~55.51) | 0.001* |
| Dmean-GTV (Gy) | 73.89(72.62~75.27) | 74.44(72.04~75.30) | 0.981 |
| Dmean-GTVnd (Gy) | 70.77(66.09~74.87) | 72.41(68.22~73.98) | 0.235 |
| Volume-GTV (cc) | 26.49(8.37~43.04) | 29.32(11.95~46.46) | 0.649 |
| Volume-GTVnd (cc) | 8.14(0.5~17.33) | 8.04(0.63~81.05) | 0.355 |
| III | - | - | - |
| Dmean to cervical | 51.92(44.25~57.83) | 53.52(49.22~57.21) | 0.013 |
| Dmean-GTV (Gy) | 73.53(69.02~75.93) | 73.94(69.31~78.21) | 0.204 |
| Dmean-GTVnd (Gy) | 70.51(63.50~73.19) | 70.81(63.78~74.67) | 0.628 |
| Volume-GTV (cc) | 34.78(6.05~74.58) | 32.64 (6.77~93.72) | 0.583 |
| Volume-GTVnd(cc) | 7.74 (0.72~54.39) | 28.58 (1.19~49.86) | 0.712 |
| IVa | - | - | - |
| Dmean to cervical | 53.47(50.95~59.42) | 55.67(48.05~59.03) | 0.006* |
| Dmean-GTV (Gy) | 74.26 (73.20~75.63) | 73.92 (65.78~76.12) | 0.346 |
| Dmean-GTVnd (Gy) | 73.45 (69.34~74.97) | 72.51 (64.36~79.29) | 0.319 |
| Volume-GTV (cc) | 58.91 (39.74~106.54) | 42.87 (3.09~135.76) | 0.236 |
| Volume-GTVnd (cc) | 17.3 (2.48~48.67) | 28.58 (1.89~83.80) | 0.173 |
| II~IVa | - | - | - |
| Dmean to cervical | 51.88(44.25~59.42) | 54.11(48.05~59.03) | 0.000* |
| Dmean-GTV (Gy) | 73.63(69.02~75.93) | 73.94(67.87~78.21) | 0.855 |
| Dmean-GTVnd (Gy) | 71.28(63.50~74.97) | 71.47(63.78~79.29) | 0.655 |
| Volume-GTV (cc) | 34.78(6.05~106.54) | 38.00(3.09~135.76) | 0.443 |
| Volume-GTVnd(cc) | 8.36(0.5~54.39) | 10.82(0.63~83.8) | 0.019* |
| Target | Dmin (Gy) | Dmax (Gy) | Dmean (Gy) | Volume (cc) |
|---|---|---|---|---|
| PGTV | 60.00 | 70.00 | 73.77(65.87~78.21) | 39.00(3.09~135.76) |
| PGTV-nd | 61.00 | 70.00 | 70.90(63.50~79.29) | 17.74(0.50~83.80) |
| PTV1 | 54.00 | 63.00 | 68.42 (62.34~73.34) | 237.07(40.50~978.00) |
| PTV2 | 50.00 | 56.00 | 61.25(56.10~67.86) | 900.75(95.83~1651.20) |
Note: Dmin=Minimum dose of radiation.
Dmax=Maximum dose of radiation.
Dmean=Mean dose of radiation.
Cases were collected strictly per the inclusion criteria during a predefined time period: November 2018 to June 2020. Objective data, such as imaging findings, were used, and outcome assessment was performed in compliance with the RTOG.
4. RESULTS
4.1. Efficacy of Treatment
Disease assessment was conducted on 109 patients, among which 16/109(14.68%), 62/109 (56.88%), and 31/109 (28.44%) had stage II, III, and IVa disease, respectively. As of December 31, 2023, 16 patients had died. The CR, PR, and SD for the whole cohort were 81.65%, 2.75%, and 0.92%, respectively. The 1-year OS, DFS, DMFS, and LRFS were 98.17%, 86.24%, 95.41%, and 96.33%, respectively. The 3-year OS, DFS, DMFS, and LRFS were 86.24%, 81.65%, 84.40% and 83.49%, respectively (Table 5).
| End Point | Outcome |
|---|---|
| Overall survival | - |
| Rate at 1 y, % (95% CI) | 98.17 |
| Rate at 3 y, % (95% CI) | 86.24 |
| Failure-free survival | - |
| Rate at 1 y, % (95% CI) | 86.24 |
| Rate at 3 y, % (95% CI) | 81.65 |
| Distant metastasis-free survival | - |
| Rate at 1 y, % (95% CI) | 95.51 |
| Rate at 3 y, % (95% CI) | 84.40 |
| Locoregional relapse-free survival | - |
| Rate at 1 y, % (95% CI) | 96.33 |
| Rate at 3 y, % (95% CI) | 83.49 |
| Response to the whole treatment | - |
| Complete response | 89(81.65) |
| Partial response | 3(2.75) |
| Stable response | 1(0.92) |
| Death | 16(14.68) |
Note: *Calculated using the stratified Cox proportional hazards regression model.
4.2. Toxicities
Acute adverse events are listed in Table 6. Adverse events were reported by the majority of patients, with the most common ones being neutropenia and gastrointestinal tract reactions. The incidence of grade 1 and 2 neutropenia and gastrointestinal tract reaction were 72.48% and 88.1%, respectively, while grade 3 neutropenia and gastrointestinal tract reaction were observed in 27.52% and 11.9% of patients. Five patients experienced hepatoxicity and 1 developed nephrotoxicity. Overall, adverse events associated with concurrent chemoradiotherapy were generally manageable, with no reports of grade 4 neutropenia, nausea, or vomiting. RD, the most common radiation-induced complication, affected patients with grades 1, 2, 3, and 4 in 0.92%, 40.37%, 53.38%, and 7.33% of all patients, respectively. Moderate to severe RD (grades 3 and 4) was observed in 60.71% of participants.
| Event | Grade 1 or 2 | Grade 3 | Grade4 |
|---|---|---|---|
| Radiation dermatitis | 45(41.28%) | 56(51.38%) | 8(7.34%) |
| Neutropenia | 79(72.48%) | 30(27.52%) | 0 |
| Nausea and Vomiting | 96(88.1%) | 13(11.9%) | 0 |
| Hepatoxicity | 5(4.59%) | 0 | 0 |
| Nephrotoxicity | 1(0.92) | 0 | 0 |
In the univariate analysis, node stage and TMN stage demonstrated significant associations with an increased likelihood of grade 3/4 RD. Dosimetric factors, including Dmean to the cervical site and volume of GTV-nd, were analyzed for their relationship with RD severity. Subsequently, multivariate analysis identified Dmean to the cervical site as an independent predictor (OR=1.003; 95%CI=1.001-1.005; P=0.003).
We assessed the Dmean to the neck in 109 patients, ranging from 4425 cGy to 5942 cGy, with an average of 5330 cGy. To clarify in detail the toxicity levels of radiation-induced skin injury associated with different radiation doses, patients were categorized into four groups based on Dmean: <5000 cGy (group 1, n = 9), 5001-5200 cGy (group 2, n = 25), 5201-5500 cGy (group 3, n = 51), and >5500cGy (group 4, n = 24). We compared the incidence of RD between mild (grade 1 and 2) and moderate/severe (grade 3 and 4) groups across these Dmean categories: 6 vs. 3 (mild vs moderate/severe) cases in group 1, 18 vs. 7 cases in group 2, 17 vs. 34 cases in group 3, and 4 vs. 20 cases in group 4 (Table 7). As anticipated, there were significantly more cases of severe RD in higher Dmean groups (group 2 vs. 3, P=0.03; group 2 vs. 4, P<0.01), with no statistical significance between groups 3 and 4. Further stratification into ≤5200 cGy (N=34) and >5200 cGy (N=76) groups revealed a notably higher incidence of grade 3 or 4 RD in the high Dmean group (Dmean>5200 cGy, RD grade≤2 vs. RD grade≥3, P<0.01). Receiver operating characteristic (ROC) curve analysis indicated that a Dmean >52.06 Gy was predictive of grade ≥3 RD (AUC=0.733%; P<0.01; 95%CI= 0.637-0.829) (Figs. 1-3).
| Radiothemities | Grade 1 | Grade 2 | Grade 3 | Grade 4 | Grade 1+2 | Grade 3+4 | P-value |
|---|---|---|---|---|---|---|---|
| <5000cGy | 0 | 6 | 3 | 0 | 3 | 6 | 0.763 |
| 5000~5200cGy | 0 | 18 | 6 | 1 | 7 | 18 | 0.003 |
| 5201~5500cGy | 1 | 16 | 29 | 5 | 34 | 17 | 0.173 |
| >5500cGy | 0 | 4 | 18 | 2 | 20 | 4 | - |
| <5200cGy | 0 | 24 | 9 | 1 | 10 | 24 | - |
| ≥5200cGy | 1 | 20 | 47 | 7 | 54 | 21 | 0.000* |
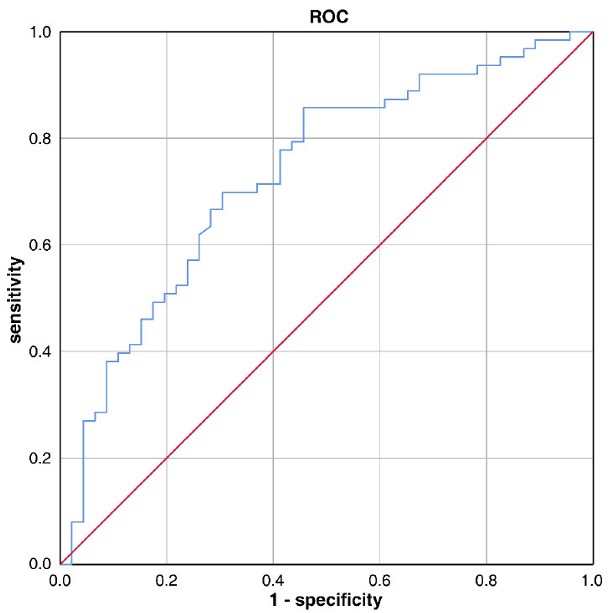
Receiver operating characteristic curves of grade≥3 radiation dermatitis according to mean cervical dose in all participants(n=109). Optimal cut-off mean dose of cervical was 5206cGy (AUC=0.733%;p<0.01;95%CI=0.637~0.829).
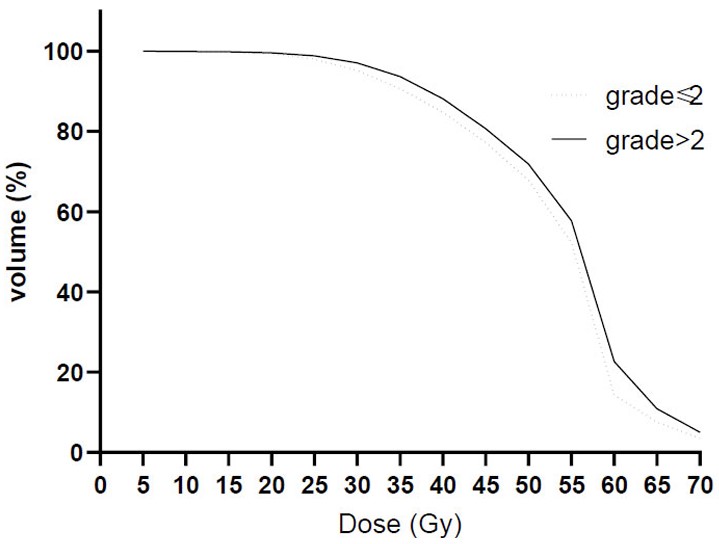
Averaged dose-volume histograms for patients with grade≤2 (dashed) and grade>2 (solid) radiation dermatitis.
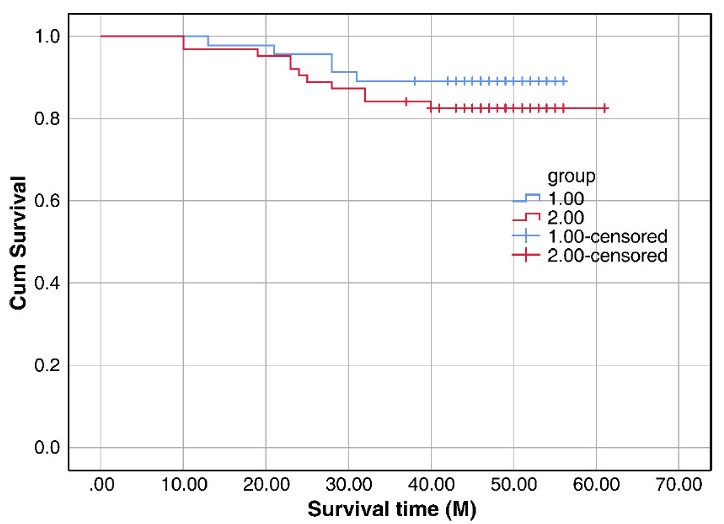
Kaplan-Meier analysis of survival curves of overall survival (OS).
5. DISCUSSION
Radiation dermatitis, ranging from erythema to severe desquamation, results from epithelial cell depletion due to radiation exposure [11], and is assessed according to the RTOG criteria for severity. It is reported that 41.4% of head and neck cancer patients suffer grade 3 or 4 dermatitis [12], if untreated, can lead to severe complications, even death. Our investigation highlights the development of severe acute radiation dermatitis in patients undergoing concurrent chemoradiotherapy, correlating with specific dosimetric factors. By comparing dermatitis incidence across different mean neck dose groups, we identified that a mean cervical dose exceeding 5200 cGy may induce severe dermatitis. A prospective clinical study [12] on acute radiotherapy side effects in nasopharyngeal carcinoma demonstrated that advanced radiation-focusing techniques can prevent skin irradiation and reduce skin damage. However, the study did not address factors such as radiotherapy dose and target volume. Bonomo P [12] concluded that the volume of the 2-mm skin ring receiving 5000 cGy and 6000 cGy as the optimal dosimetric predictors, with corresponding values of 19.9 cc and 5.8 cc, respectively (AUC = 0.61 for both). The development framework based on the dose-toxicity surface histogram (DTH) enables severity-based classification and predictive dose assessment of RD, with a predicted dose of 5430 cGy for Grade 3 RD [12]. This study reviewed and summarized parameters in radiation plans for patients with nasopharyngeal carcinoma, with a specific focus on analyzing cervical skin doses. This approach allowed for a more direct and comprehensive assessment of the correlation between radiation dermatitis development and cervical radiation exposure. We found that Dmean=5206 cGy was highly consistent with the findings of previous studies. In a case study reported by Donghayun Kim [13], the mean dose to level I-VII neck nodes was found to be 5860 cGy, further validating the reliability and clinical relevance of the results in the present study.
The mechanism of RD remains unclear, and standardized treatments for this condition are lacking. Preventive interventions can significantly reduce RD-related damage, with optimized radiotherapy dose serving as a key measure to mitigate RD [14, 15]. While prior studies have established an association between cervical radiation dose and dermatitis severity [16], detailed dose-specific data regarding RDremain scarce. This study aims to clarify in detail the toxicity levels of such skin damage across different radiation doses and provides precise dosimetric insights. Future efforts will focus on reducing cervical radiation exposure and exploring dermatitis management strategies from existing studies.
The toxicity of RDvaries based on factors such as total radiation dose, fractionation schedule, and volume of organ or tissue treated [17]. Strategies aimed at reducing doses to the cervical site include minimizing doses to GTV-nd and CTV, as well as optimizing target volumes. Some studies have shown that elective neck irradiation can mitigate RD and enhance the quality of life [18], particularly for patients with N0-N1 nasopharyngeal carcinoma. However, this approach lacks robust, high-level evidence [19], partly due to reported in-field failures (64%) as the primary pattern of regional recurrence [20, 21]. Safely defining target volume boundaries remains crucial. Metastasis from nasopharyngeal carcinoma typically follows a predictable path through cervical lymph node levels, necessitating adequate target dose delivery for efficacy [22, 23]. Consequently, directly reducing prescription doses and volumes to the cervical site to minimize RD remains a topic of controversy.
Reducing unnecessary target volumes is a potential method to lower cervical site doses. Radiation therapy affects not only cancer cells but also nearby tissues and organs, emphasizing the importance of optimizing treatment delivery to balance tumor control with sparing normal tissues [24]. Incorporating PET/CT imaging into IMRT planning for nasopharyngeal carcinoma offers improved precision in delineating GTVnd volumes and elective nodal targets, enhancing treatment accuracy, uniformity, and efficiency [25].
Efforts to mitigate cervical site radiation doses remain limited, necessitating a comprehensive review of clinical approaches to reduce skin toxicity. Variations in radiosensitivity and toxicity profiles across different chemotherapy regimens are often overlooked in current chemoradiotherapy protocols [26]. Specifically, higher incidences of acute grade 3 skin toxicity have been associated with 5-fluorouracil use [27], suggesting avoidance of this agent during radiotherapy. Radioresistance poses a significant challenge in managing nasopharyngeal carcinoma [28], prompting exploration into combination therapies to potentially overcome this issue. Notably, immunotherapy, particularly in cases with high PD-L1 expression, offers a promising avenue for enhancing treatment efficacy [29], despite potential immune-related adverse events affecting the skin and appendages [30]. Clinical trials evaluating anti-PD-L1 antibodies and other immune checkpoint inhibitors in various treatment contexts are ongoing, with further investigation needed on their impact on radiation dermatitis [31].
While establishing definitive guidelines for minimizing cervical site dose and volume remains challenging, clinicians can implement interventions to manage skin toxicity during chemoradiotherapy. Topical corticosteroids reduce the production of inflammatory mediators, modulate inflammatory/immune responses, cell proliferation, and vasoconstriction, thereby lowering the risk of RD [32, 33]. An antiulcer oral mucosal protectant creates a microenvironment that promotes the self-healing of damaged mucosal epithelial cells, enabling the prevention and treatment of oral mucositis and mucosal ulcers [34]. Epidermal growth factor (EGF) maintains homeostasis by regulating the proliferation, growth, and migration of epithelial cells. It also induces angiogenesis and provides nutritional support for tissues [35]. These interventions have additionally been shown to benefit the treatment of RD. A silicone-based film-forming gel dressing, which creates a barrier to reduce mechanical friction and transepidermal water loss (TEWL), is recommended for the prevention of radiation dermatitis [36]. Advancements in radiotherapy techniques, such as TOMO therapy, show promise in improving therapeutic ratios for treating nasopharyngeal carcinoma [37]. However, further clinical data and studies are necessary to justify broader application, considering economic considerations and treatment efficacy.
6. LIMITATIONS OF THE STUDY
Given the current gaps in evidence, ongoing challenges in nasopharyngeal carcinoma treatment and clinical management, and the inherent limitations of our study in terms of sample size and duration of follow-up, future research should involve larger cohorts and long-term quality of life assessments. Such efforts are essential to advance the management of radiation dermatitis in nasopharyngeal carcinoma treatment protocols.
CONCLUSION
Based on the findings of this study, maintaining a Dmean <5200 cGy for the neck site may mitigate severe skin toxicities and delay the onset of moderate/severe radiation dermatitis. One should carefully consider reducing cervical doses to protect neck skin integrity, ensuring precise and adequate delivery to target volumes in patients with locally advanced nasopharyngeal carcinoma. Based on current evidence and literature, we recommend early topical treatments (such as EGF and corticosteroids) for patients with Dmean ≥5200 cGy during radiotherapy.
The correlation between RD and whole cervical site dose remains inadequately explored, with no consensus on optimal radiation doses for this region in nasopharyngeal carcinoma patients. Our research contributes to understanding the clinical characteristics of radiation dermatitis and proposes a potential strategy to mitigate skin damage.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contribution to the paper as follows: W.F.: Study conception and design; W.X.: Data collection; W.L.: Data Analysis or Interpretation; W.J.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| CCRT | = Concurrent chemoradiotherapy |
| Dmean | = Mean dose |
| RFFS | = Regional failure-free survival |
| CR | = Complete remission |
| PR | = Partial remission |
| SD | = Stable disease |
| OS | = Overall survival |
| DFS | = Disease-free survival |
| DMFS | = Distant metastasis-free survival |
| LRFS | = Locoregional relapse-free survival |
| RTOG | = Radiation therapy oncology group |
| RD | = Radiation dermatitis |
| NPC | = Nasopharyngeal carcinoma |
| AJCC | = American joint committee on cancer |
| KPS | = Karnofsky |
| MRI | = Magnetic resonance imaging |
| CT | = Computed tomography |
| ECT | = Emission computed tomography |
| EBV-DNA | = Epstein-barr virus-deoxyribonucleic acid |
| IMRT | = Intensity-modulated radiation therapy |
| VMAT | = Volumetric-modulated arc therapy |
| NCCN | = National comprehensive cancer network |
| GTV | = Gross tumor volume |
| CTV | = Clinical target volume |
| DVH | = Dose-volume histograms |
| PET/CT | = Positron emission tomography/computed tomography |
| CI | = Confidence interval |
| ROC | = Receiver operating characteristic |
| AUC | = Area under curve |
| DTH | = Dose-toxicity surface histogram |
| TEWL | = Transepidermal water loss |
| PD-L1 | = Programmed death-1 ligand |
| TOMO | = Tomotherapy |
| EGF | = Epidermal growth factor |
| NA | = Not applicable |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Ethics Committee of Anhui Provincial Cancer Hospital, Anhui Province, China (2024-FLK-02).
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
The privacy and personal identifying information of the participants were strictly protected. Objectively, obtaining informed consent from the participants was impracticable. Furthermore, this research project involved no commercial interests. For these reasons, written informed consent from the patients was not required for this study.
AVAILABILITY OF DATA AND MATERIALS
The datasets used and the analysis during the current study are available from the corresponding author on reasonable request.
ACKNOWLEDGEMENTS
We acknowledge the support of the Department of Radiotherapy for Oncology, Anhui Cancer Hospital.


