All published articles of this journal are available on ScienceDirect.
Atrial Pseudoaneurysm Secondary to Pacemaker Implant Complicated with Device Infection and Pericardial Abscess Formation: A Case Report
Abstract
Introduction/Background
A cardiovascular implantable electronic device infection (CIEDI) is a rare but serious complication, often difficult to diagnose. We present a unique case of CIEDI involving a long-standing right atrial pseudoaneurysm with pericardial abscess related to an old pacemaker system.
Case Presentation
A 51-year-old woman with a dual-chamber pacemaker presented with prolonged fever and lead dysfunction. Multimodal imaging revealed lead perforation into the pericardial sac, resulting in an abscess. A hybrid approach combining open surgery, percutaneous extraction, and targeted antibiotic therapy led to full recovery.
Conclusion
Accurate diagnosis through advanced imaging and multidisciplinary care is essential for the management of complex CIEDI and to improve outcomes.
1. INTRODUCTION
Cardiovascular implantable electronic device infections (CIEDI) represent a serious and potentially life-threatening complication. The diagnosis is often challenging due to nonspecific symptoms and limitations of conventional imaging. Pseudoaneurysms related to device leads have been described in various anatomical locations. To our knowledge, this is the first reported CIEDI case involving an old right atrial (RA) pseudoaneurysm, associated with previous lead perforation. Such rare structural complications require prompt recognition and multidisciplinary management. This report highlights the critical role of advanced multimodal imaging and combined surgical and medical treatment to achieve complete recovery in managing complex CIEDI.
2. CASE PRESENTATION
A 51-year-old female patient with a 23-year history of a left-sided cephalic dual-chamber pacemaker implantation, performed at another institution for unknown reasons (the patient reports “for syncope with pauses”), presented to the emergency department with fever and constitutional symptoms that had been ongoing for the past two months. She reported daily fevers reaching up to 38°C, along with intermittent epigastric discomfort and left shoulder pain.
The patient had undergone a generator replacement three years earlier, without evidence of infection at the generator site, and recalled experiencing chest pain for several days following implantation of the original system 30 years ago.
Physical examination was unremarkable, without cardiac murmurs, lymphadenopathy, or skin lesions. The electrocardiogram showed a paced ventricular rhythm with retrograde atrial conduction. Chest X-ray (Fig. 1A, B) demonstrated appropriately positioned pacemaker leads within the right heart chambers. The only significant laboratory abnormality was an elevated C-reactive protein level of 7.5 mg/dL, without leukocytosis or neutrophilia. Blood cultures and multiple serological tests were obtained as part of the fever of unknown origin workup, all of which returned negative results.
The patient was subsequently admitted to the Internal Medicine Department, and a contrast-enhanced computed tomography (CT) of the body was performed. The scan revealed that the atrial lead of the pacemaker traversed the inferoposterior wall of the RA. The lead tip appeared to be located within the pericardial sac, surrounded by a centimeter-sized fluid collection suggestive of abscess formation (Fig. 2A-D). The vena cavae were dilated, and the pericardium demonstrated mild enhancement and effusion suggestive of probable pericarditis.
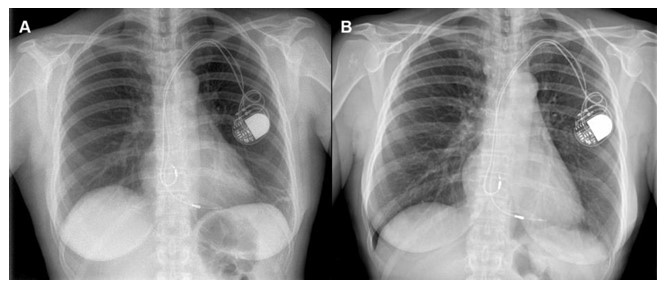
Chest X-rays showing apparently normal positioning of pacemaker leads after the generator replacement performed three years earlier (A) and currently (B), with an almost imperceptible difference in the positioning of the atrial lead tip.
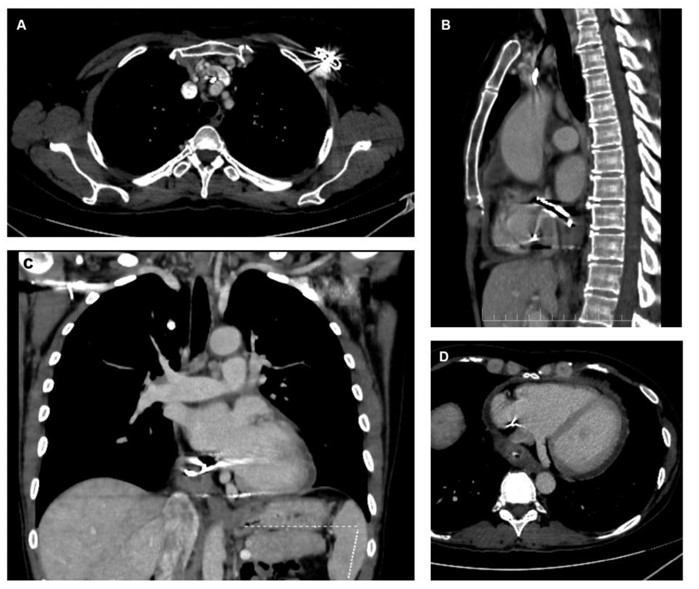
Contrast-enhanced CT scan showing (A) axial view of the pacemaker generator area with venous thrombus adjacent to the leads, (B) sagittal view with extravascular lead trajectory, (C) coronal view of the abscessed area in the pericardial sac, and (D) axial view showing the relationship of the abscess to the coronary sinus and inferior vena cava.
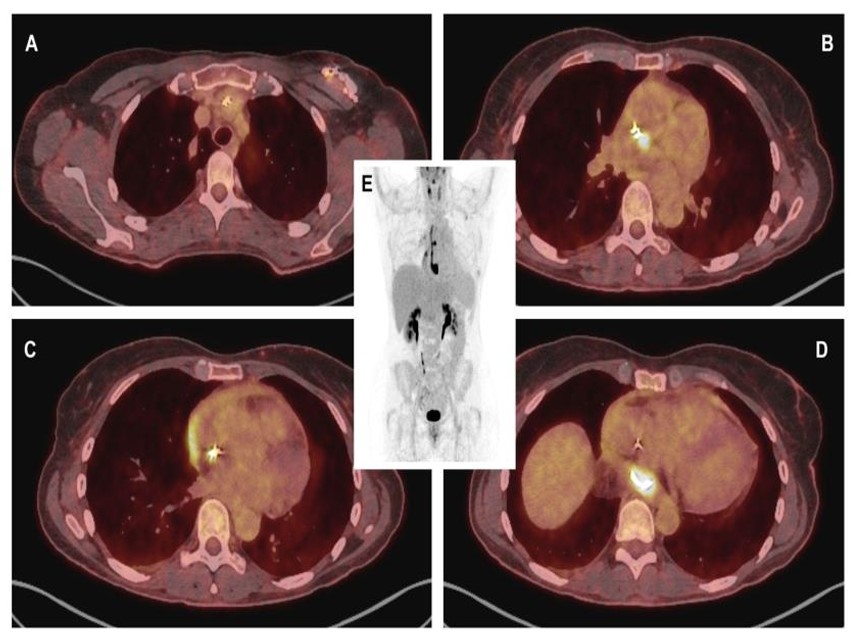
Fused CT-PET with 18F-FDG images (A) showing slight metabolic activity at the lead exit point, with more intense uptake from the entrance into the innominate vein extending to its distal end; (B) exhibiting greater focality in the middle third; and (D) demonstrating the region of the paraesophageal pericardial abscess, with a maximum standardized uptake value (SUVmax) of 16. (C) A thin line of activity was observed in the pericardial effusion, more intense in the region adjacent to the right atrium. (E) Volumetric PET reconstruction demonstrates the previously described findings.
Repeat blood cultures were obtained, and empiric antibiotic therapy with daptomycin and meropenem was initiated. Transthoracic echocardiography (TTE) showed no valvular involvement, with normal biventricular size and function. An anechoic cavity was observed in the inferoposterior wall of the RA, posterior to the superior vena cava (SVC), containing the lead tip. A mild adjacent pericardial effusion was also noted, along with significant acceleration of SVC flow (Supplementary Videos1-4). Very late and mild filling of the cavity was observed following echocardiographic contrast administration (Supplementary Video5). Transesophageal echocardiography (TEE) confirmed the absence of valvular involvement.
Positron emission tomography with 18F-fluorodeoxyglucose (18F-FDG PET) was performed, which confirmed the suspected diagnosis, demonstrating infection of the generator and leads with involvement of the atrial wall and formation of a pericardial abscess (Fig. 3A-E). Both multiplex FilmArray testing and blood cultures remained negative after 14 days.
During pacemaker interrogation, an atrial lead exhibiting reduced impedance, undersensing, and a markedly elevated stimulation threshold was identified, consistent with previous recordings. Additionally, the patient was not pacemaker-dependent, maintaining an adequate sinus rhythm and atrioventricular conduction.
The case was discussed in a multidisciplinary session, and open surgery was decided upon. Through two left pectoral incisions, the electrodes were mobilized along their intravenous course to the RA using a Cook release system and Biomenco EZ expandable guides, along with removal of the generator and its pocket, and debridement of surrounding tissues (Fig. 4A-F). Via the median longitudinal sternotomy, the RA and SVC were partially opened, allowing removal of the RA lead, which was embedded in the atrial wall up to the entry point of the abscess. An old pseudoaneurysm with a small inlet between the inferior vena cava and the coronary sinus was identified. A large amount of adherent thrombotic material was also removed along with the leads. The abscess cavity was left open to the RA, and the atrial incision was closed with a double 4/0 Prolene suture. Samples from the leads and other locations were sent for microbiological analysis. Drains and a temporary epicardial pacemaker were left in place for three days and subsequently removed, given the absence of conduction abnormalities.
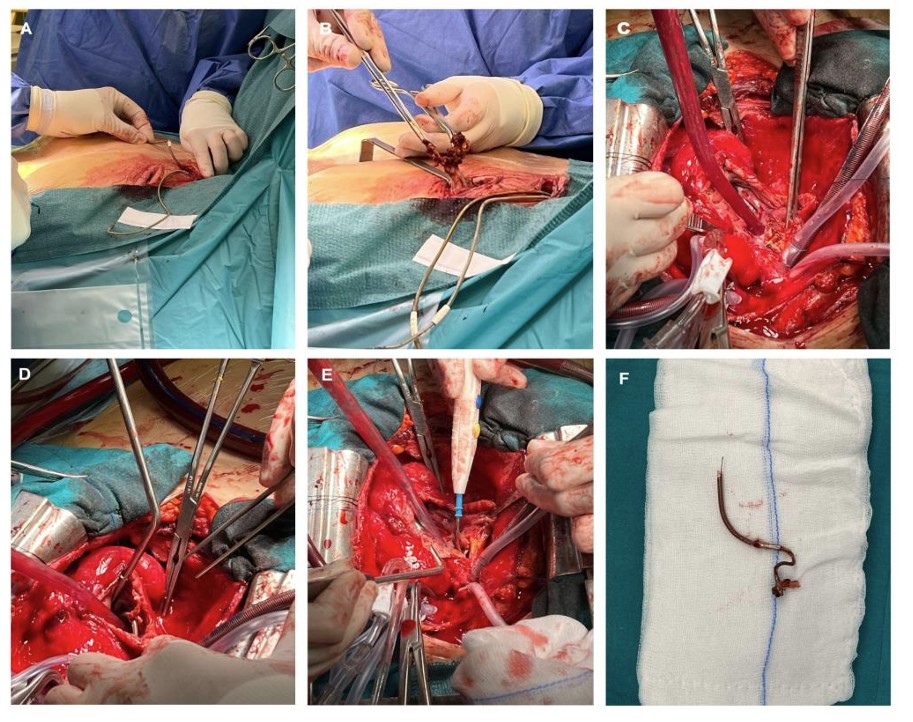
Surgical mechanical release of (A) the electrodes along their vascular path to the RA and ventricle using the Cook release system and Biomenco EZ expandable guides and (B) pacemaker pocket appearance after release of the generator and electrodes. (C) After electrode release, a median sternotomy was performed, followed by dissection of pericardial adhesions, heparinization, and cannulation for initiation of cardiopulmonary bypass (arterial cannula inserted into the ascending aorta, venous cannulas inserted in the SVC and right femoral vein). The distal SVC wall and RA were opened, revealing the entry of the electrode into the pseudoaneurysm/abscess area between the inferior vena cava and the coronary sinus. (D) Dissection and complete release of the remaining atrial electrode. (E) Drainage of the abscessed area, left open to the RA. (F) Atrial electrode after release.
Cloxacillin-resistant Staphylococcus epidermidis was isolated from the atrial lead culture. Meropenem was then discontinued, and daptomycin continued for two weeks. Its isolation was confirmed via polymerase chain reaction (PCR) targeting the 16S subunit of bacterial ribosomal ribonucleic acid (rRNA). After a follow-up TTE showed no abnormalities, oral linezolid was started for an additional four weeks.
Anticoagulation was initiated following the diagnosis of right internal jugular vein thrombosis, which resolved during follow-up. The patient was asymptomatic and discharged sixteen days after the procedure, showing a favorable clinical course without relapse and with negative follow-up blood cultures.
3. DISCUSSION
This case has illustrated a CIEDI with abscess formation of a probably ancient atrial pseudoaneurysm secondary to pacemaker lead wall perforation, extended to the rest of the device and associated with localized pericarditis and septic venous thrombosis.
Cases of right ventricular [1, 2] and SVC pseudoaneurysms [3] have previously been reported, usually related to device lead implantation and explantation procedures, percutaneous structural interventions, and cardiac surgeries. However, we have not found in the literature any reported case of RA pseudoaneurysm after pacemaker implantation. We believe it remained undiagnosed for nearly three decades, considering both the chest pain reported by the patient after implantation and its location, which is difficult to detect using the commonly employed techniques (TTE and CT).
Considering the causative agent, it is likely that CIEDI was derived from an unidentified source of infection, such as a simple blood draw with secondary bacteremia. Nevertheless, it remains conceivable that it originated from the generator replacement three years earlier, manifesting a particularly slow progression and minimal clinical expression.
CIEDI occurs in 1-7% of implanted devices and is complicated by infectious endocarditis in up to 10-27% of cases. Repeated interventions in pacemaker systems are considered the greatest isolated risk factor for CIEDI. Renal and cardiac failure, the use of corticosteroids and anticoagulants, and diabetes are also recognized as risk factors [4].
The presentation may be acute (occurring up to six weeks after implantation) or chronic, with the former being more clinically expressive and less difficult to diagnose. The endocarditic process is characterized by recurrent bacteremia and systemic clinical symptoms, rheumatic disorders, or symptoms secondary to distant endocarditic embolization. In generator infections, local inflammatory signs may also be present [5].
Although rare, as described in other cases, the device dysfunction may raise diagnostic suspicion [6]. In this case, the atrial lead had been dysfunctional since the earliest available recordings, with a relative worsening over the past few months. The laboratory findings of CIEDI are usually minimal and non-specific, occasionally leading to elevated acute phase reactants [4].
The microorganisms most frequently involved in CIEDI are coagulase-negative Staphylococci, especially in chronic infections. Obtaining blood cultures prior to initiating antibiotic therapy, as well as samples from the device after removal, is essential not only for the diagnosis of CIEDI but also for tailoring the antibiotic regimen based on susceptibility testing. Additionally, the use of new molecular techniques (e.g., 16S bacterial rRNA PCR) allows diagnosis in a higher percentage of cases [7, 8].
Echocardiography, due to its low invasiveness, is useful for ruling out complications that may alter the therapeutic plan, keeping in mind that the presence of vegetations on device leads does not confirm a diagnosis of CIEDI, nor does their absence exclude it, since these may represent non-infective thrombotic formations without clinical significance. TEE has greater sensitivity than TTE, allowing a more thorough assessment of structures, such as the RA and SVC. Finally, 3D echocardiographic reconstructions provide improved spatial visualization of anatomical structures [9, 10].
If, as in this case, there are no external signs of infection in the generator or microbiological isolation, 18F-FDG CT-PET scanning is recommended by international guidelines as the ideal and most sensitive tool for identifying CIEDI. It is also useful for detecting potential primary sources of infection or distant infectious embolisms [11, 12].
Generally, complete removal of the device is recommended once the diagnosis has been established, preferably through percutaneous methods. If this is not feasible, as in this patient, open surgery may be performed, either alone or combined with extraction systems that facilitate removal and prevent embolization of infectious material [4, 13, 14].
Antibiotic therapy should be considered adjunctive, and it is recommended that empirical therapy be initiated via the parenteral route and continued for at least 2 weeks after the device removal in cases with documented bacteremia, and for up to 4-6 weeks in complicated cases [5, 15].
The indication for reimplantation should be carefully reassessed, considering whether the original indication persists and balancing the potential risks and benefits. In our patient, this was ultimately deemed unnecessary, with her syncope at a young age attributed to an excessive cardioinhibitory neuro-mediated response [1, 13, 15].
CONCLUSION
Although clinical findings were nonspecific, persistent high fever in a patient with a pacemaker prompted the diagnosis of complicated CIEDI, which carried potentially life-threatening implications. The combination of multimodal imaging techniques provided valuable information on the origin and extent of the infection, enabling improved therapeutic planning. Communication between specialists was essential for the positive outcome, achieved through a multidisciplinary approach in a condition otherwise associated with a high risk of mortality.
AUTHORS’ CONTRIBUTIONS
It is hereby acknowledged that all authors have accepted responsibility for the manuscript's content and consented to its submission. They have meticulously reviewed all results and unanimously approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| 18F-FDG PET | = Positron emission tomography with 18F-fluorodeoxyglucose |
| rRNA | = Ribosomal ribonucleic acid |
| CIEDI | = Cardiovascular implantable electronic device infection |
| CT | = Computed tomography |
| PCR | = Polymerase chain reaction |
| RA | = Right atrium |
| SUVmax | = Maximum standardized uptake value |
| SVC | = Superior vena cava |
| TEE | = Transesophageal echocardiography |
| TTE | = Transthoracic echocardiograph |
HUMAN AND ANIMAL RIGHTS
All clinical investigations performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee, and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
Verbal informed consent was obtained from the patient for inclusion in this case report and for publication of anonymized clinical data and imaging.
AVAILABILITY OF DATA AND MATERIALS
All the data and supporting information are provided within the article.
ACKNOWLEDGEMENTS
Declared none.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publisher’s website along with the published article.
SUPPLEMENTARY MATERIAL LEGEND
Supplementary Video 1
Biplanar transthoracic echocardiographic image in the left parasternal plane, showing the right ventricular inflow tract in transverse and longitudinal views, with a pulsatile formation in the inferoposterior wall of the right atrium, close to the opening of the vena cavae.
Supplementary Video 2
Biplanar transthoracic echocardiographic image in the parasternal plane, as shown in Supplementary Video 1, demonstrating color Doppler flow acceleration at the inflow of the vena cavae.
Supplementary Video 3
Four-chamber apical echocardiographic view centered on the right chambers, showing a pulsatile pseudoaneurysm in the inferoposterior wall of the right atrium and systolic-diastolic flow with color Doppler inside of it, as well as its communication with the right atrium.
Supplementary Video 4
Echocardiographic 3D reconstruction from apical four-chamber view centered on the right chambers, showing a pseudoaneurysm and an anechoic collection in the pericardial sac, with the tip of the pacemaker lead visible inside.
Supplementary Video 5
Four-chamber apical echocardiographic view centered on the right chambers showing late filling of the atrial pseudoaneurysm 30 seconds after administration of the echocardiographic contrast.


