All published articles of this journal are available on ScienceDirect.
Case Report: Eosinophilic Myocarditis as a Paraneoplastic Syndrome in Classical Hodgkin’s Lymphoma
Abstract
Introduction
Eosinophilic myocarditis is a rare and aggressive form of infiltrative cardiomyopathy associated with significant morbidity and mortality, especially when diagnosis is delayed. Symptoms are often vague and overlap with other cardiac conditions, making recognition challenging. Positron emission tomography combined with computed tomography has become more relevant as a supplementary diagnostic tool, although its effectiveness in follow-up remains underexplored. This report discusses a likely paraneoplastic case in classical Hodgkin’s lymphoma, emphasizing that metabolic cardiac activity on serial imaging closely mirrored clinical progression.
Case Presentation
A 75-year-old woman with classical Hodgkin’s lymphoma initially responded well to chemotherapy and maintained stability for over eighteen months. She later developed progressive heart failure with restrictive physiology, biventricular involvement, severe pulmonary hypertension, and an apical intraventricular thrombus. Laboratory tests indicated intermittent eosinophilia and varying inflammatory markers. Multimodal imaging revealed diffuse myocardial edema and extensive endocardial fibrosis, indicating eosinophilic involvement. Across five oncological scans, myocardial metabolic activity increased alongside the patient's deterioration. Despite thorough investigations, including biopsies and ruling out infectious, autoimmune, infiltrative, and coronary causes, histological confirmation remained elusive. Given her advanced cardiac condition, comorbidities, and complications from treatment, immunosuppression and invasive interventions were not viable options, and the patient continued to decline.
Conclusion
This case illustrates the diagnostic challenges of eosinophilic myocarditis in unusual paraneoplastic contexts and underscores the value of multimodal imaging and interdisciplinary collaboration. The metabolic changes observed across sequential scans support this approach for early detection and follow-up, though additional validation is required.
1. INTRODUCTION
Eosinophilic Myocarditis (EM) is a rare and aggressive form of infiltrative cardiomyopathy that is frequently linked to poor outcomes. Diagnosing it can be difficult due to nonspecific symptoms and its overlap with other cardiac conditions. Positron Emission Tomography-Computed Tomography (PET-CT) has become a valuable tool not only for diagnosis but also for monitoring disease progression, even though its role in follow-up is not well documented. We present a case of paraneoplastic EM in a patient with classical Hodgkin’s Lymphoma (cHL), showcasing the innovative use of PET-CT for ongoing assessment.
2. CLINICAL CASE
A 75-year-old woman with no notable past medical issues was diagnosed in July 2021 with cHL, stage IIB, and some concerning prognostic features. She received two cycles of chemotherapy using the ABVD regimen (adriamycin, bleomycin, vinblastine, and dacarbazine). A positive early response was noted on an interim fluorine-18 fluorodeoxyglucose (18f-FDG) PET-CT at the end of 2021, leading to a reduction in therapy to an additional 3.5 cycles of AVD. A complete metabolic response was observed on PET-CT in 2022, and the patient remained symptom-free for the next 18 months.
At the end of 2023, relapse was suspected due to constitutional symptoms, and scattered hypermetabolic adenopathies were identified on PET-CT. A lymph node biopsy revealed only reactive changes. The patient also experienced flu-like symptoms for several weeks, including a persistent cough and diarrhea, with normal colonoscopy findings (no pathological specimens were obtained).
In early 2024, the patient was admitted to the Cardiology Department with a first episode of Heart Failure (HF), reporting progressive dyspnea and bilateral ankle edema over the preceding weeks. N-terminal pro-brain natriuretic peptide (NT-ProBNP) was significantly elevated at 9,000 pg/mL, and renal function was mildly impaired. Acute-phase reactants were mildly elevated (high-sensitivity C-reactive protein [hs-CRP], 36 mg/L), and the patient presented with relative eosinophilia (absolute eosinophil count of 800 cells/µl, accounting for 13% of total leukocytes). Her previously unremarkable electrocardiogram now showed sinus tachycardia with a “pseudo-Q” pattern in leads V1 to V4, as well as secondary repolarization abnormalities suggestive of Left Ventricular (LV) strain (Fig. 1). The patient reported intermittent precordial discomfort, with mildly elevated high-sensitivity troponin T (levels ranging from 20 to 40 ng/L).
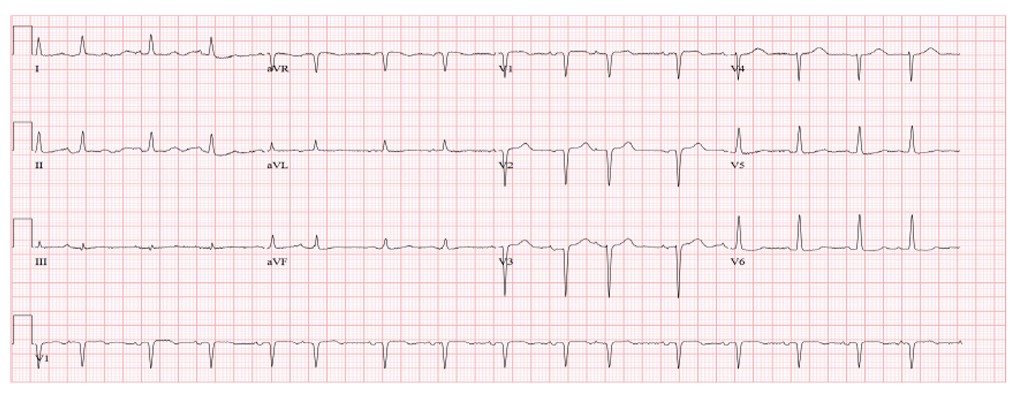
Electrocardiogram showing sinus tachycardia at 130 bpm with “pseudo-Q” waves in leads V1 to V4 and secondary repolarization abnormalities, consistent with left ventricular strain.
The Transthoracic Echocardiogram (TTE), which was normal prior to chemotherapy, now showed reduced LV volumes, severe diastolic restriction, and apical obliteration due to an intraventricular thrombus. Systolic function remained intact, with no obvious regional wall motion issues. However, global longitudinal strain was significantly reduced (at -11%), predominantly impacting the apical segments (Fig. 2). The Right Ventricle (RV) appeared dilated with decreased systolic function. Significant Mitral Regurgitation (MR) and very severe pulmonary hypertension were suspected (estimated systolic pulmonary artery pressure, 105 mmHg) (Supp. videos 1-3). Depletive therapy and oral anticoagulation were initiated.
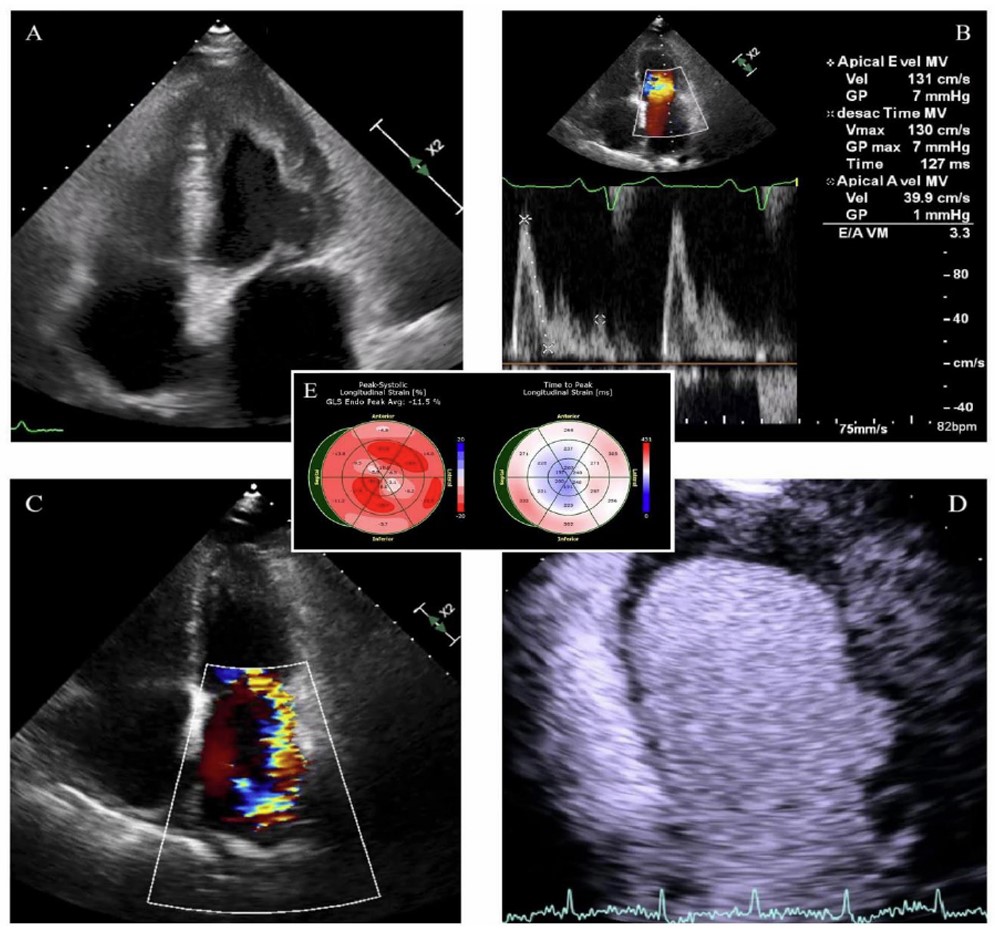
Transthoracic echocardiography, apical four-chamber view, showing: (A) a left ventricle with reduced volumes secondary to extensive apical obliteration by an intraventricular thrombus; (B) a transmitral filling pattern consistent with grade III diastolic dysfunction; (C) significant mitral regurgitation with right ventricular dilation and biatrial enlargement; (D) an obliterative apical thrombus with low enhancement following ultrasound contrast administration, associated with a basal inferolateral aneurysm; (E) significantly compromised GLS (at -11%), predominantly affecting the apical segments, with a relatively increased time-to-peak at the basal level, supporting the hypothesis of apical endomyocardial dysfunction or structural impairment, as well as impaired conduction involving the basal segments.
The study was completed with Cardiac Magnetic Resonance (CMR) imaging, which additionally revealed myocardial edema and extensive endocardial fibrosis in the LV, both diffuse and predominantly located in the mid-apical and inferolateral walls (Fig. 3).
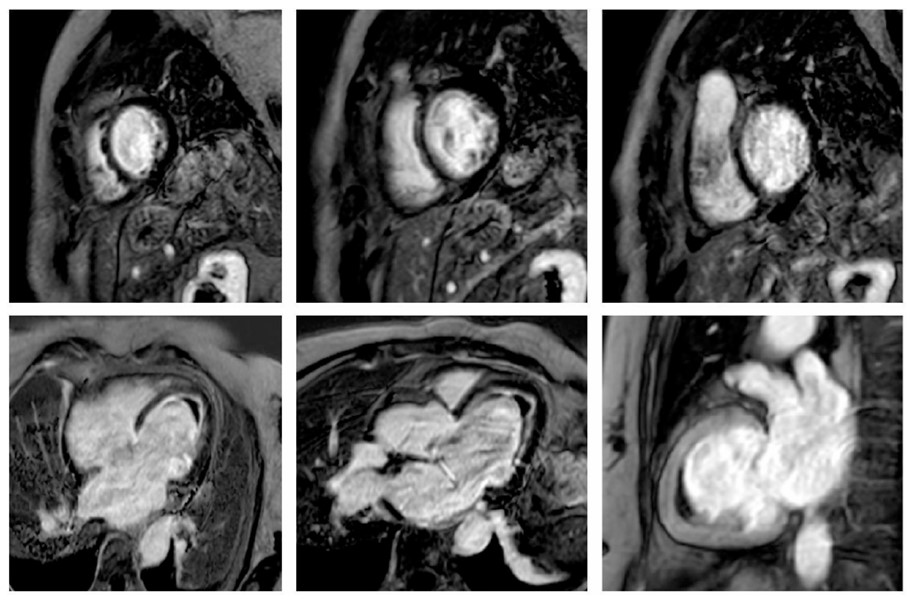
Cardiac magnetic resonance T1-weighted sequences following gadolinium-based contrast administration demonstrate diffuse ring-like endomyocardial enhancement in the left ventricle, predominantly involving the mid and apical segments, as well as the basal inferolateral wall. These findings suggest extensive endocardial fibrosis, apical obliteration due to intraventricular thrombus, and incipient basal aneurysm formation. Extensive myocardial edema is also evident on the final T2-weighted sequence. Taken together, these findings are highly indicative of active eosinophilic myocarditis.
Given these findings, cardiac amyloidosis was initially suspected but subsequently ruled out through cardiac scintigraphy, hematologic workup, and biopsies of subcutaneous fat and the right ventricular septal endomyocardium. However, the myocardial biopsy did not yield additional etiologic information regarding the patient’s infiltrative cardiomyopathy (Fig. 4). Flow cytometry at that time excluded hematologic malignancies in peripheral blood, except for the previously noted mild eosinophilia.
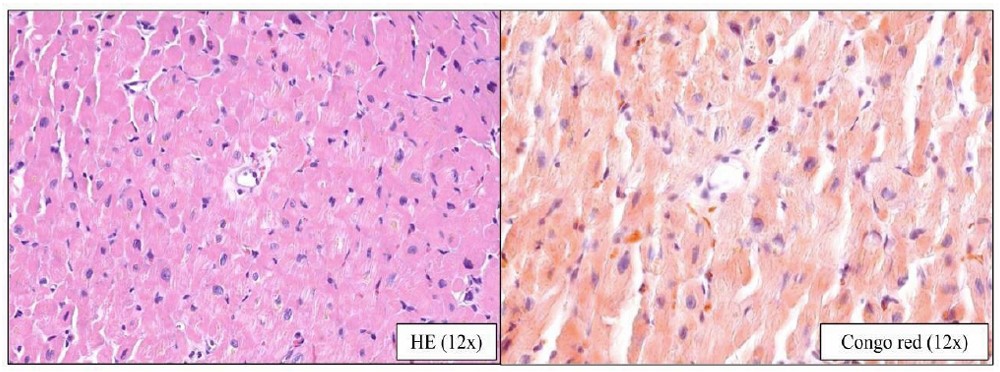
Histopathological specimen at 12x magnification from the endomyocardial biopsy, obtained from the septal region of the right ventricle: (A) Hematoxylin–eosin staining showing myocardial tissue without fibrosis or obvious eosinophilic infiltrates; (B) Congo red staining demonstrating the absence of apple-green birefringence under polarized light. These findings are non-diagnostic and limited to the sampled region; therefore, they do not exclude infiltration in non-biopsied myocardial areas (e.g., the left ventricle).
Right heart catheterization confirmed severe mixed pre- and post-capillary pulmonary hypertension, with a reduced cardiac index of 2.3 L/min/m2. Ventilation–perfusion scintigraphy excluded chronic pulmonary thromboembolism, and coronary angiography ruled out coronary artery disease while revealing angiographically severe MR (Supp. video 4). Infectious causes, including viral and parasitic infections, were ruled out, and the autoimmune panel was unremarkable. Although the exact etiology of the restrictive cardiomyopathy could not be definitively established at that time, it was attributed to probable anthracycline (doxorubicin)-induced cardiotoxicity, given a cumulative dose of 275 mg/m2.
The patient was referred to our center eight months after diagnosis for evaluation of interventional options. Surgical mitral valve replacement was deemed futile, and edge-to-edge repair was ruled out due to technical limitations. Heart transplantation was contraindicated because of her age, pulmonary hypertension, and a recent, poor-prognosis lymphoma. LV assist device implantation was excluded due to overt RV dysfunction (Supp. video 5). Empirical immunosuppressive treatment was dismissed because of significant infectious complications and the advanced stage of her cardiopathy. Lastly, palliative medical management with diuretics and neurohormonal therapy was chosen, although the patient showed poor tolerance to these treatments. With progressive clinical deterioration and development of refractory HF, the patient died only ten months after diagnosis.
A retrospective case review revealed peaks in acute-phase reactants, eosinophilia, and β2-microglobulinemia prior to chemotherapy, with transient normalization following tumor response and a subsequent rise at the time of cardiac disease onset (Fig. 5). In five consecutive PET-CT scans conducted to monitor her oncologic disease, signs indicating myocardial inflammation were observed, aligning with spikes in laboratory markers (Fig. 6). Taken together with the highly suggestive cardiac CMR findings, these results support a probable diagnosis of paraneoplastic EM known in its thrombotic phase as Loeffler endocarditis, within the context of cHL, and likely worsened by doxorubicin-based chemotherapy.
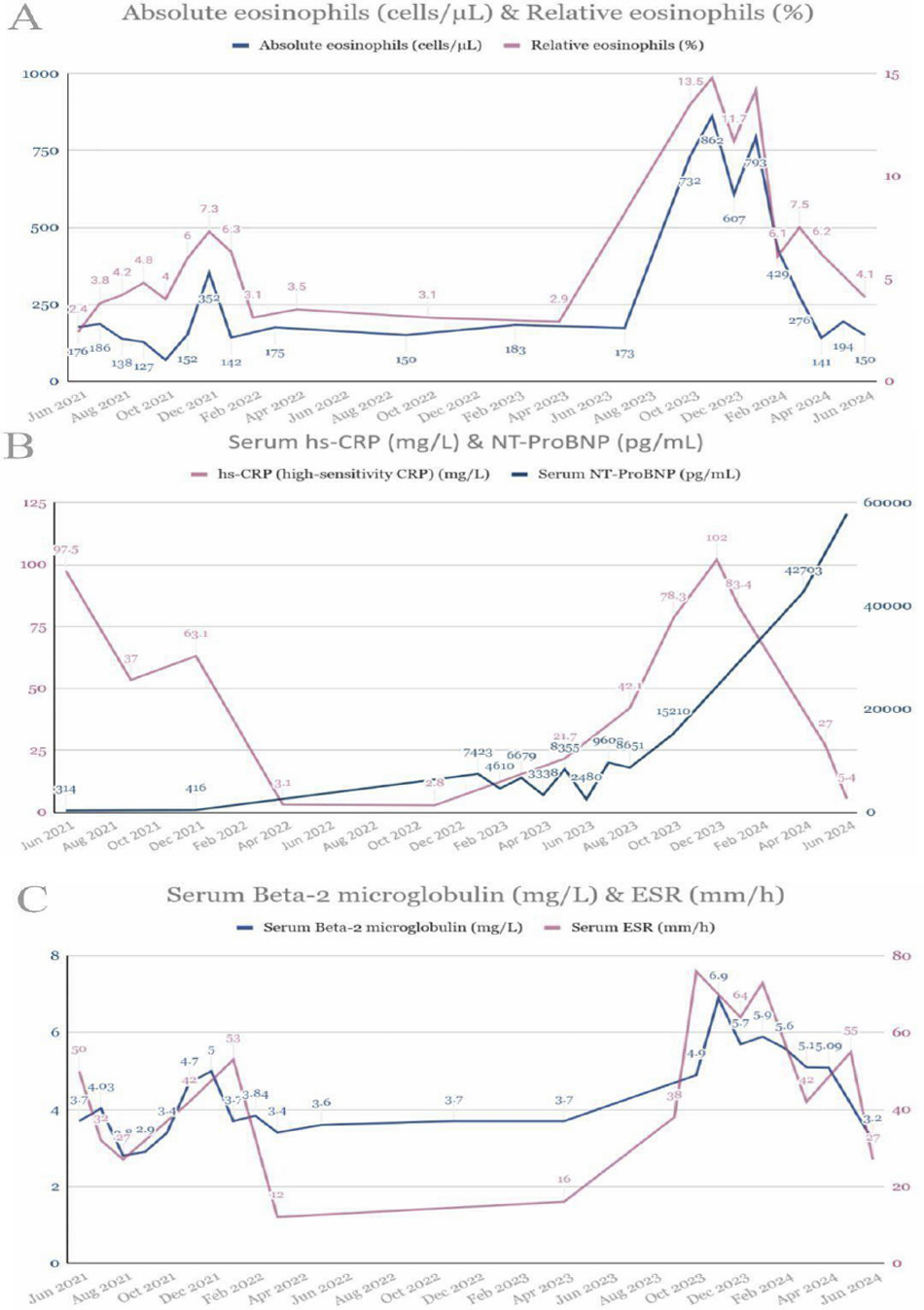
Dot plot graphs showing temporal variation in the following parameters: (A) absolute and relative eosinophilia; (B) serum levels of hs-CRP and NT-ProBNP; and (C) serum β2-microglobulin and erythrocyte sedimentation rate (ESR). Both eosinophilia and the various acute-phase reactants exhibit a bimodal distribution, with an initial peak during the active phase of the oncological condition, followed by a temporary normalization after chemotherapy, and a second increase preceding the clinical onset of cardiomyopathy. Natriuretic peptide levels, which were initially normal, gradually rose after this second peak, indicating the accelerated progression of cardiomyopathy to more advanced stages.
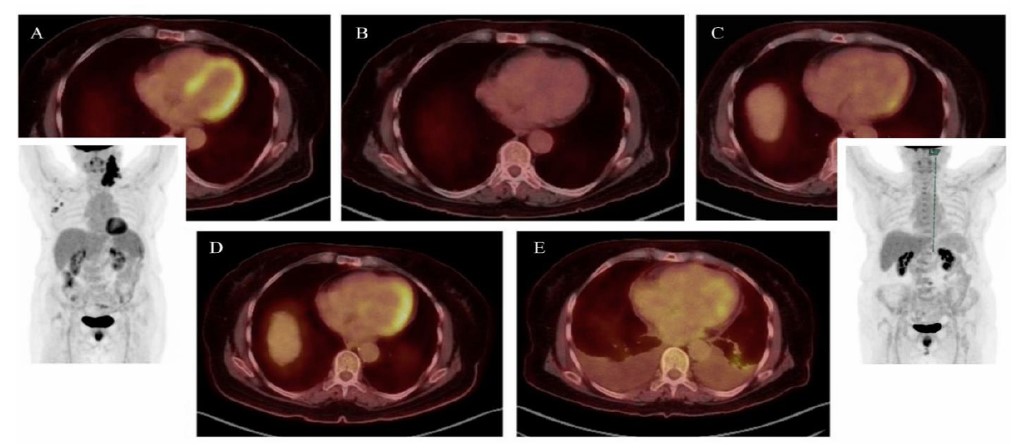
Sequential PET-CT axial and volumetric reconstructions during patient follow-up, demonstrating: (A) the active phase of Hodgkin’s lymphoma with prominent, widespread myocardial uptake (late 2021); (B) following initial chemotherapy cycles, a complete oncologic response with no myocardial uptake (early 2022); (C) several months after finishing chemotherapy, revealing early signs of myocardial uptake while the patient remained asymptomatic from a cardiovascular perspective (late 2022); (D) the onset of clinical heart failure, exhibiting intense, asymmetric uptake corresponding to areas of increased gadolinium enhancement on CMR (mid 2023); (E) end-stage cardiomyopathy, showing relatively decreased and patchy uptake, consistent with established myocardial fibrosis (mid 2024, two months before the patient’s passing).
3. DISCUSSION
Despite its diagnostic difficulties, EM is a rare yet highly aggressive form of restrictive cardiomyopathy associated with a poor prognosis, particularly if not identified in its early stages. The differential diagnosis includes tumor infiltration, reactions from radiation or drugs, infections, genetic disorders, amyloidosis, sarcoidosis, and various autoimmune diseases. Treatment primarily involves corticosteroids and other immunosuppressants, with a favorable response possible if started promptly [1].
The coexistence of cHL and peripheral eosinophilia has been well established and is thought to stem from tumor cell production of various cytokines and growth factors. Some studies suggest that up to 60% of patients with lasting hypereosinophilia (>1500 cells/µL) exhibit symptoms of EM [2]. However, there are instances where patients with normal or only slightly elevated eosinophil counts have been documented in oncohematologic cases, particularly those with cHL. This indicates eosinophilic translocation with secondary organ damage, likely due to heightened tissue sensitivity, cellular dysregulation, and other factors [3]. A bone marrow biopsy is a useful method for evaluating eosinophilic tissue infiltration, ruling out monoclonal hematologic disorders, and identifying potential gene mutations, all of which can assist in determining targeted treatment options [4].
Doxorubicin-based chemotherapy, a well-known cardiotoxic agent and a recognized contributor of diffuse endocardial fibrosis (typically with more subtle and patchy involvement that goes beyond the endocardium) [5], may have played a role in the patient's condition. As highlighted by the 2022 ESC guidelines, cancer patients undergoing chemotherapy are likely to benefit from structured monitoring strategies to identify early signs of cardiotoxicity through regular testing [6]. In this case, EM-related metabolic activity observed on sequential PET-CT scans with unprecedented temporal correlation underscores the potential of this imaging modality for both early diagnosis and longitudinal follow-up [7].
An RV septal endomyocardial biopsy ruled out other potential causes of infiltrative heart disease; however, this procedure has a sensitivity as low as 65% for EM. Since up to 40% of EM cases may show isolated LV involvement, image-guided LV biopsy was considered but ultimately dismissed due to the high risk of thromboembolic events. Some authors suggest that a diagnosis and subsequent initiation of immunosuppressive therapy could have been warranted based on highly suggestive CMR findings within a paraneoplastic context, even when endomyocardial biopsy results were inconclusive or absent [8]. However, this approach was not pursued in our patient for the reasons outlined above.
CONCLUSION
EM remains a rare and poorly understood infiltrative cardiomyopathy, linked to significant morbidity and mortality, especially if targeted treatment is not promptly initiated. Diagnosing it is a multidisciplinary challenge that involves ruling out various pathologies and relies significantly on laboratory testing, multimodal imaging, and effective interdisciplinary collaboration. A typical paraneoplastic presentation can complicate the diagnostic process, making cooperation among specialists crucial to prevent rapid and irreversible decline. In this context, our case report suggests that PET-CT may be a valuable complementary tool for initial assessment in patients with a high clinical suspicion, although further validation and long-term studies are needed to confirm its diagnostic and prognostic significance.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contributions to the paper as follows: M.J.R.I., A.M.M., A.S.P., I.D.M.M., and F.A.G.N.: Prepared the draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| ABVD | = Adriamycin, Bleomycin, Vinblastine, Dacarbazine |
| cHL | = Classical Hodgkin’s Lymphoma |
| CMR | = Cardiac Magnetic Resonance |
| EM | = Eosinophilic Myocarditis |
| ESR | = Erythrocyte Sedimentation Rate |
| FDG | = Fluorodeoxyglucose |
| GLS | = Global Longitudinal Strain |
| HF | = Heart Failure |
| hs-CRP | = High Sensitivity C-Reactive Protein |
| LV | = Left Ventricle / Left Ventricular |
| MR | = Mitral Regurgitation |
| NT-ProBNP | = N-Terminal Pro-Brain Natriuretic Peptide |
| PET-CT | = Positron Emission Tomography–Computed Tomography |
| RV | = Right Ventricle / Right Ventricular |
| TTE | = Transthoracic Echocardiogram |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable, as this case report includes no identifiable personal data and all images are fully anonymized.
HUMAN AND ANIMAL RIGHTS
All clinical investigations performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or research committee, and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
Verbal informed consent was obtained from the patient for participation in this report and for the publication of anonymized clinical data and imaging.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this published article.
ACKNOWLEDGEMENTS
All individuals listed as authors have contributed substantially to the performance and reporting of the work.
AI DECLARATIONS
The English translation of this manuscript was supported by generative Artificial Intelligence (AI), specifically ChatGPT (OpenAI, GPT-5.1 model), which helped improve translation quality and clarity.
SUPPLEMENTARY MATERIALS
Supplementary material is available on the publisher’s website along with the published article.
Supplementary video 1: Transthoracic echocardiography, apical four-chamber view, showing a left ventricle with preserved systolic function, an obliterative apical thrombus, and a basal inferolateral aneurysm with restricted motion of the mitral valve leaflets; a mildly dilated right ventricle with mildly reduced systolic function, and significant biatrial enlargement.
Supplementary video 2: Echocardiography, apical four-chamber view after ultrasound contrast administration, showing an extensive apical thrombus with low enhancement in the left ventricle, along with a basal inferolateral aneurysm.
Supplementary video 3: Transesophageal echocardiography, mid-esophageal view at 145º, showing an inferobasal aneurysm and left atrial and mitral annular dilation, with restricted motion of both mitral valve leaflets—more pronounced in the posterior leaflet, which is also short (19 mm). Failure of coaptation at the P1–A1 level results in a regurgitant jet directed toward the posterior wall, reaching the atrial roof, with systolic flow reversal in the left superior pulmonary vein, consistent with severe mitral regurgitation.
Supplementary video 4: Left ventricular angiography showing preserved ejection fraction, apical obliteration due to a laminar thrombus, a small inferobasal aneurysm, and significant mitral regurgitation with severe left atrial dilation and systolic flow reversal in the pulmonary veins.
Supplementary video 5: Transthoracic echocardiography, apical four-chamber view, showing a dilated left ventricle with mildly reduced systolic function and a residual obliterative apical thrombus; a severely dilated and dysfunctional right ventricle; and significant biatrial enlargement, more pronounced on the left.


